Abstract
Background: Many patients with type 2 diabetes mellitus (T2DM) struggle to control their glucose levels with dipeptidyl peptidase-4 inhibitor (DPP4i)-based therapy, highlighting the need to explore alternative treatments. This study aims to investigate the benefits of switching to a sulfonylurea/metformin combination in individuals with type 2 diabetes (T2D) who were previously on DPP4i. Methods: The study is a retrospective, multicenter, observational, case-based questionnaire survey conducted in T2DM patients who received DPP4i earlier but due to poor glycemic control switched to the combination in any strength. Statistical analysis was conducted using SPSS® Version 23.0 software. Continuous variables were analyzed using mean and standard deviations; categorical variables were analyzed using Fisher's exact and Chi-square tests. Results: The study analyzed data from 2,736 T2DM patients who were 18 years and above, having an average age of 38.46 ± 7.21 and average body mass index (BMI) of 27.79 ± 4.25 kg/m2. The mean change in the glycated hemoglobin (HbA1c) values after treatment was found to be 1.11 ± 0.78, while the mean change in fasting plasma glucose (FPG) and postprandial glucose (PPG) was 41.77 ± 31.11 and 67.39 ± 51.57, respectively; 94.8% of patients had no hypoglycemic events and 96.2% did not gain weight after switching to glimepiride/metformin, additionally the HbA1c, FPG, and PPG levels were control well. HbA1c before treatment was 9.64 ± 1.79 and after treatment was 7.52 ± 1.97. Similarly, FPG was 175.14 ± 89.89 mg/dL before treatment, which reduced to 133.37 ± 43.59 mg/dL after treatment. PPG was found to be 251.38 ± 80.30 mg/dL before treatment and 183.98 ± 54.76 mg/dL after treatment. Vildagliptin (50%) was the most common DPP4i being prescribed, followed by sitagliptin (30.2%). The main reason of switching to glimepiride/metformin was to improve the HbA1c levels, followed by controlling the uncontrolled glycemic levels and further improving FPG and PPG levels. Conclusion: The study supports the effectiveness and safety of switching to modern sulfonylureas/metformin in T2DM patients who are inadequately controlled on DPP4i-based therapies.
Keywords: Type 2 diabetes mellitus, DPP4i, retrospective studies, sulfonylurea, glimepiride, metformin, glycemic control
Introduction
Type 2 diabetes (T2D) is a progressive disease which often requires treatments to be added or switched in order to achieve glycated hemoglobin (HbA1c) targets. However, stringent glycemic targets in people over 65 years may increase the risk of hypoglycemia. People with multimorbidity may be less likely to receive multiple T2D therapies due to concerns about polypharmacy and drug interactions. T2D medication may be switched due to lack of efficacy or adverse drug events (ADEs)1-4.
Dipeptidyl peptidase-4 inhibitors (DPP4i) have emerged as a novel class of oral antidiabetic agents, offering glucose-lowering effects by inhibiting the breakdown of incretin hormones. However, clinical reality sometimes presents challenges as some patients fail to achieve desired glycemic control despite DPP4i therapy5.
Poor glycemic control increases the risk of cardiovascular disease, chronic kidney disease (CKD), and mortality. Studies have also shown that the use of dipeptidyl peptidase-4 (DPP-4) is contraindicated in people with CKD and might increase the risk of heart failure and acute pancreatitis6,7.
Recent studies suggest that DPP4i are associated with an increased risk of developing bullous pemphigoid (BP) in patients with diabetes8.
In many instances, discontinuation of DPP4i was found to be possible adverse events or tolerability issues related to adding insulin (58.9%), lack of efficacy/treatment goals not being met (55.4%) and cost of DPP4i in addition to insulin (48.5%)9.
Modern antidiabetic strategies have evolved to incorporate a combination of medications to target multiple facets of glucose regulation, such as modern sulfonylureas (SUs) and metformin. Modern SUs are considered ideal options due to their high efficacy, relative cardiovascular safety, and low cost.
Hence, in the pursuit of refining diabetes management strategies, the transition from DPP4i-based treatments to contemporary modern SU/metformin combinations has emerged as a potential solution for patients encountering inadequate glycemic control.
This retrospective questionnaire based study examines the outcomes and implications of such a transition, shedding light on its effectiveness and relevance in optimizing the care of patients previously uncontrolled on DPP4i therapies.
Material and Methods
Study Design
This was a retrospective, multicenter, observational, case-based questionnaire survey. It was conducted with 225 health care professionals (HCPs) across different centers in India. The study protocol was designed according to the principles of the Declaration of Helsinki.
Study Population
The study included 2,736 patients of both sexes,
aged above 18 years, diagnosed with T2DM and received DPP4i earlier but due to poor glycemic control switched to the combination in any strength. The average age of the participants was 38.46 ± 7.21 with an average body mass index (BMI) of 27.79 ± 4.25 kg/m2. Participants were also found to be overweight and obese.
Data Collection
A case report format was developed to determine the effect of switching to modern SU/metformin in patients uncontrolled on DPP4i-based therapies. The questionnaire was sent to 225 HCPs across India via an online portal. Questions regarding demographic characteristics, such as age, sex, BMI, weight change, and economic class; duration of diabetes; antidiabetic drugs used (DPP4i) and (glimepiride/metformin); weight change; hypoglycemic episodes, reasons for switching to (glimepiride/metformin); adherence to lifestyle, were included in the questionnaire. An online portal was developed where the HCPs filled in the information. A descriptive analysis was performed with the data provided on the portal.
Statistical Analysis
All continuous variables are expressed as mean ± SD (standard deviation) or median with the interquartile range per the data distribution. Categorical variables are expressed as number and their respective percentage. Differences in binary and ordinal variables between two independent groups were analyzed by the exact Chi-square test. All the reported p-values are two-sided, and p-values <0.05 are considered to indicate statistical significance. All data entries and statistical analyses were performed by using SPSS@ Version 23.0 software.
Compliance with Ethics Guidelines
The study was approved by the ethical committees at all participating centers. All procedures adhered to the ethical standards established by the relevant institutional or national research committees. Since the study used an anonymized database and was done retrospectively, patient consent was not needed.
Results
The study included, 2,736 T2DM patients who were 18 years and above with an average age of 38.46 ± 7.21. It showed significant control on the HbA1c, fasting plasma glucose (FPG), and postprandial glucose (PPG) levels after the switch. Hb1Ac before treatment was 9.64 ± 1.79 and after treatment was 7.52 ± 1.97. Similarly, FPG was 175.14 ± 89.89 mg/dL before treatment, which reduced to 133.37 ± 43.59 mg/dL after treatment. PPG was found to be 251.38 ± 80.30 mg/dL before treatment and 183.98 ± 54.76 mg/dL after treatment.
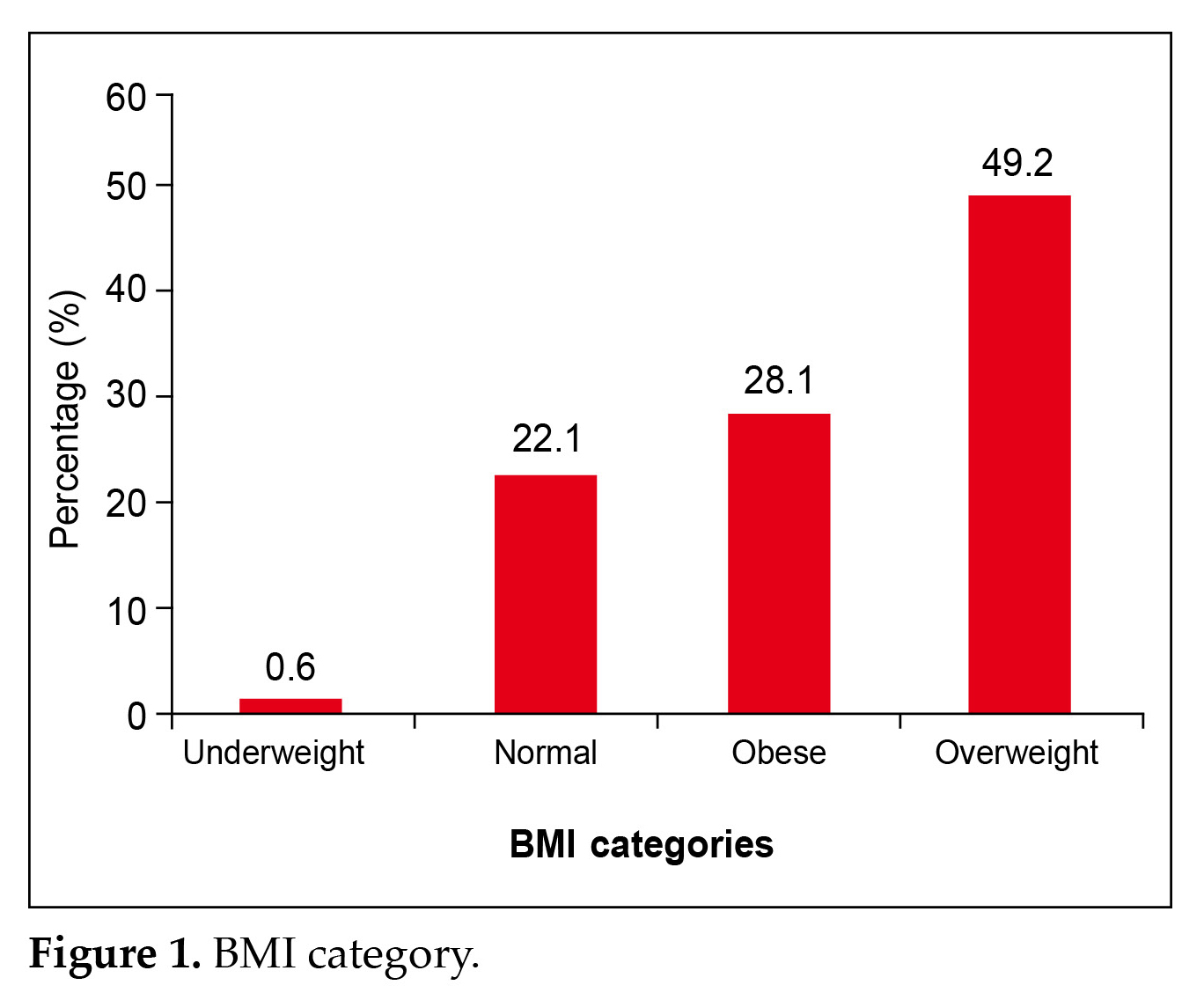
The participants had an average BMI of 27.79 ± 4.25 kg/m2; 28.1% were obese and 49.2% of the participants were overweight as shown in Figure 1.
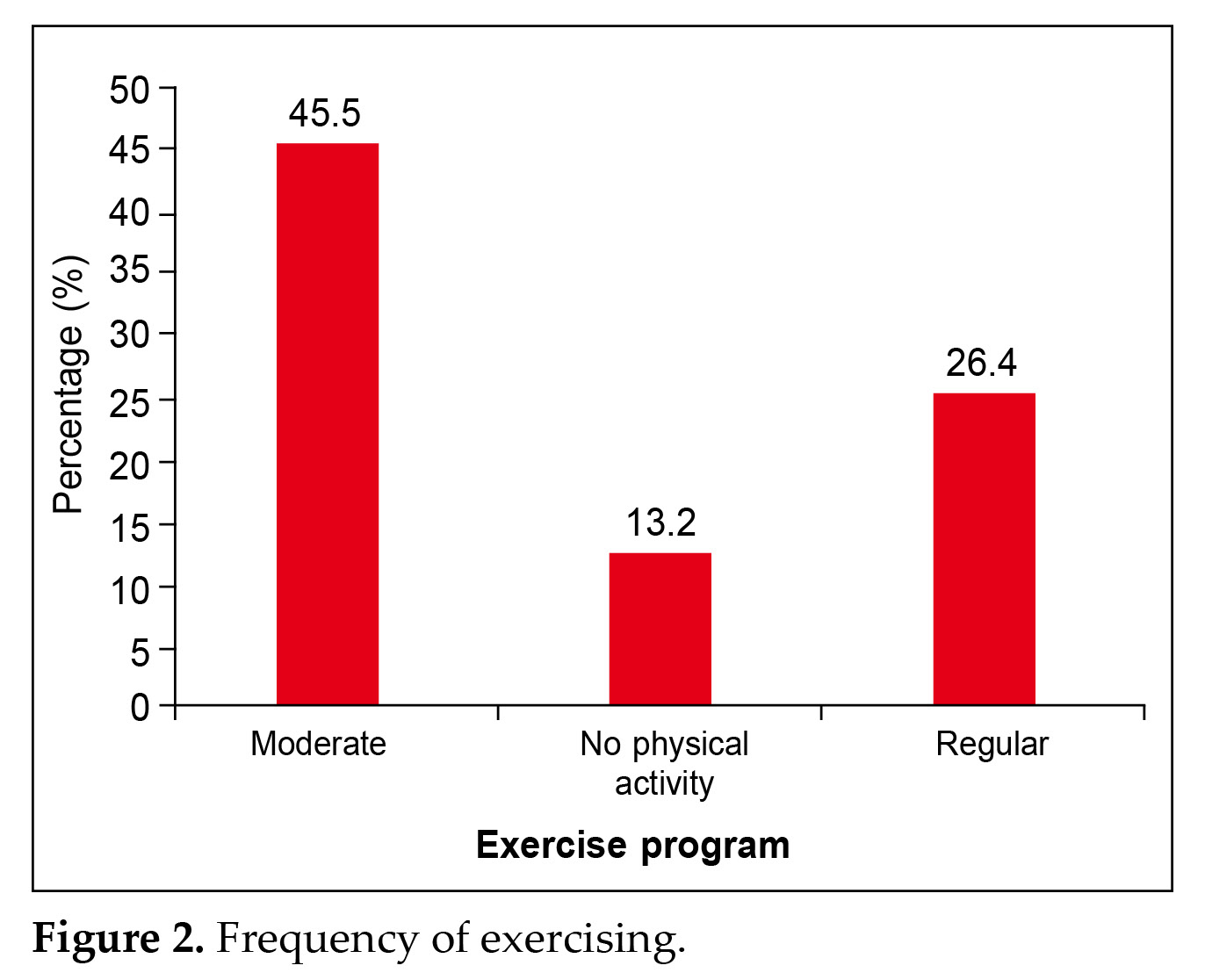
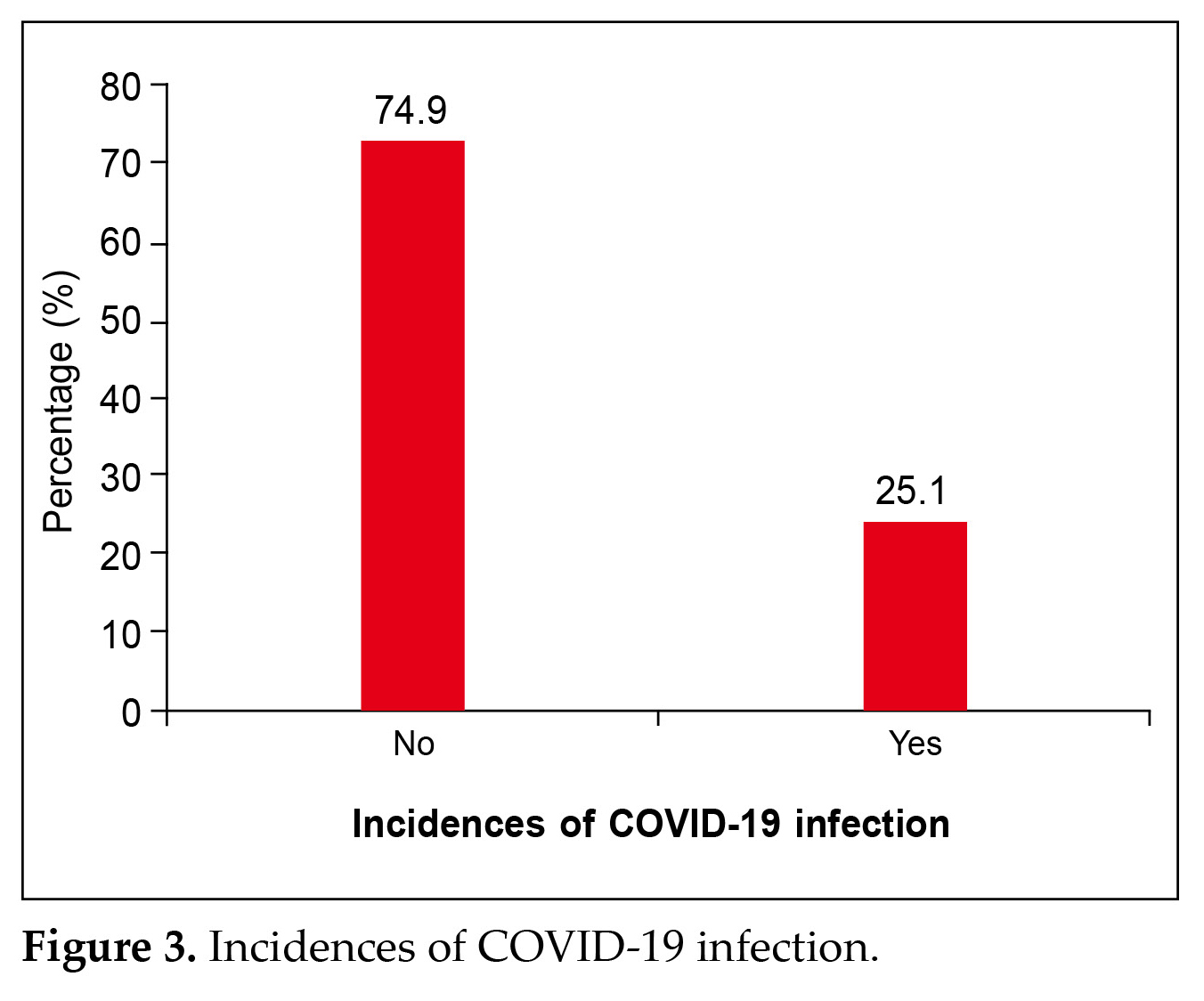
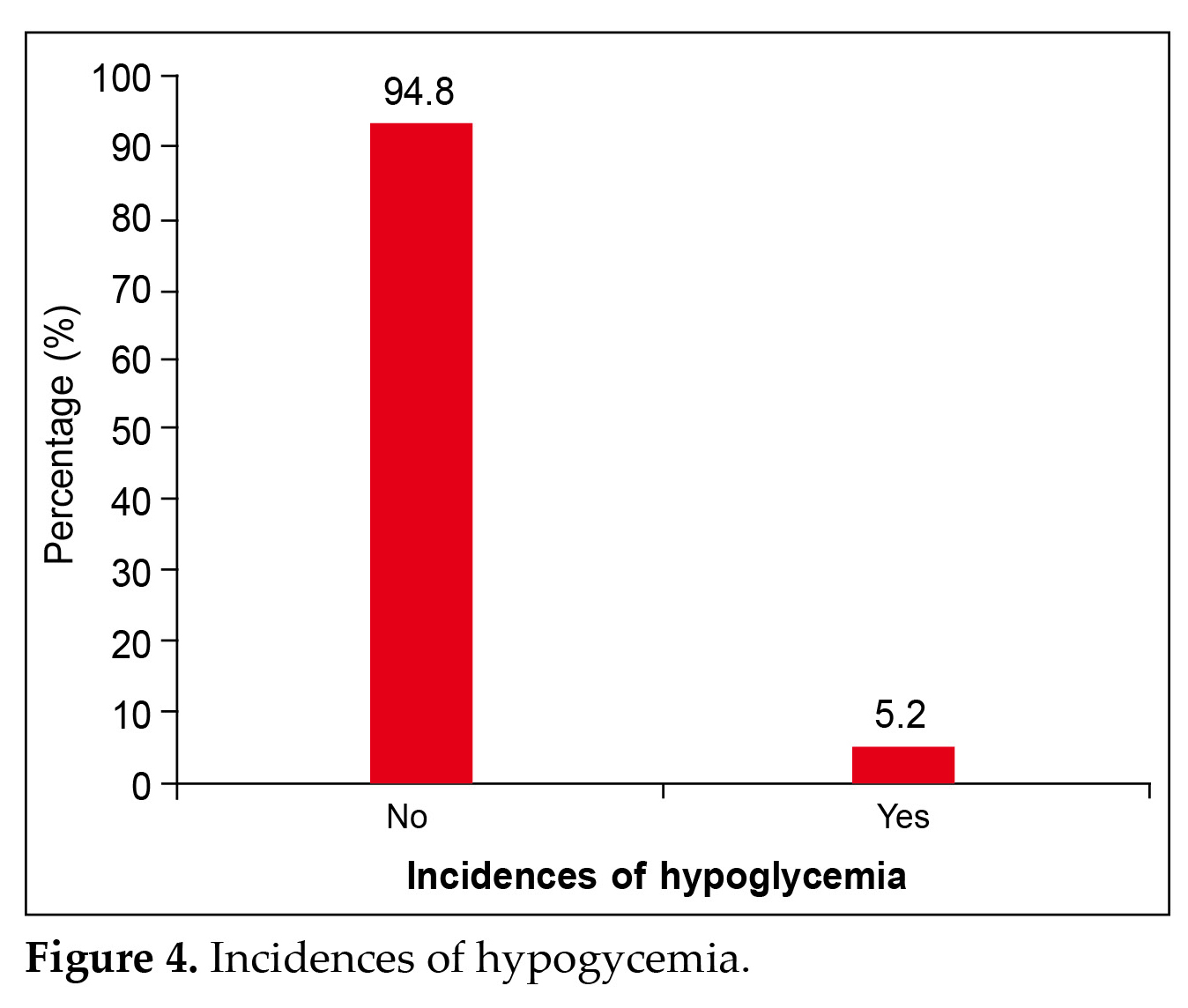
Demographic details showed that 61.5% of the participants belonged to the economically weaker section. About 45.5% were moderately active, while 26.4% were engaged in regular exercise and 13.2% were inactive as shown in Figure 2. Additionally 25.1% had a history of coronavirus disease 2019 (COVID-19) as illustrated in Figure 3. Further details showed that 94.8% of patients had no hypoglycemic events
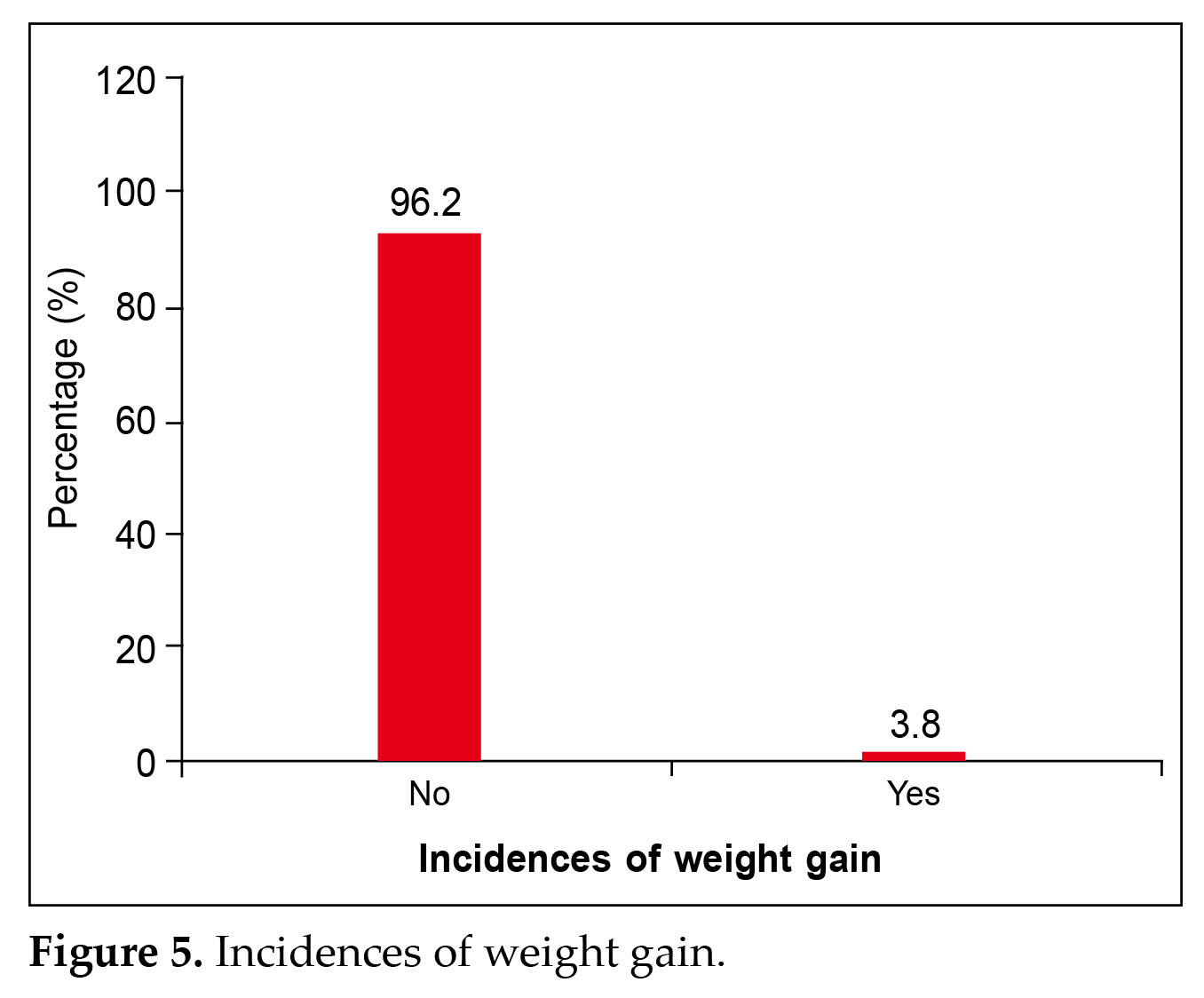
(Fig. 4) and 96.2% did not gain weight after switching to glimepiride/metformin (Fig. 5).
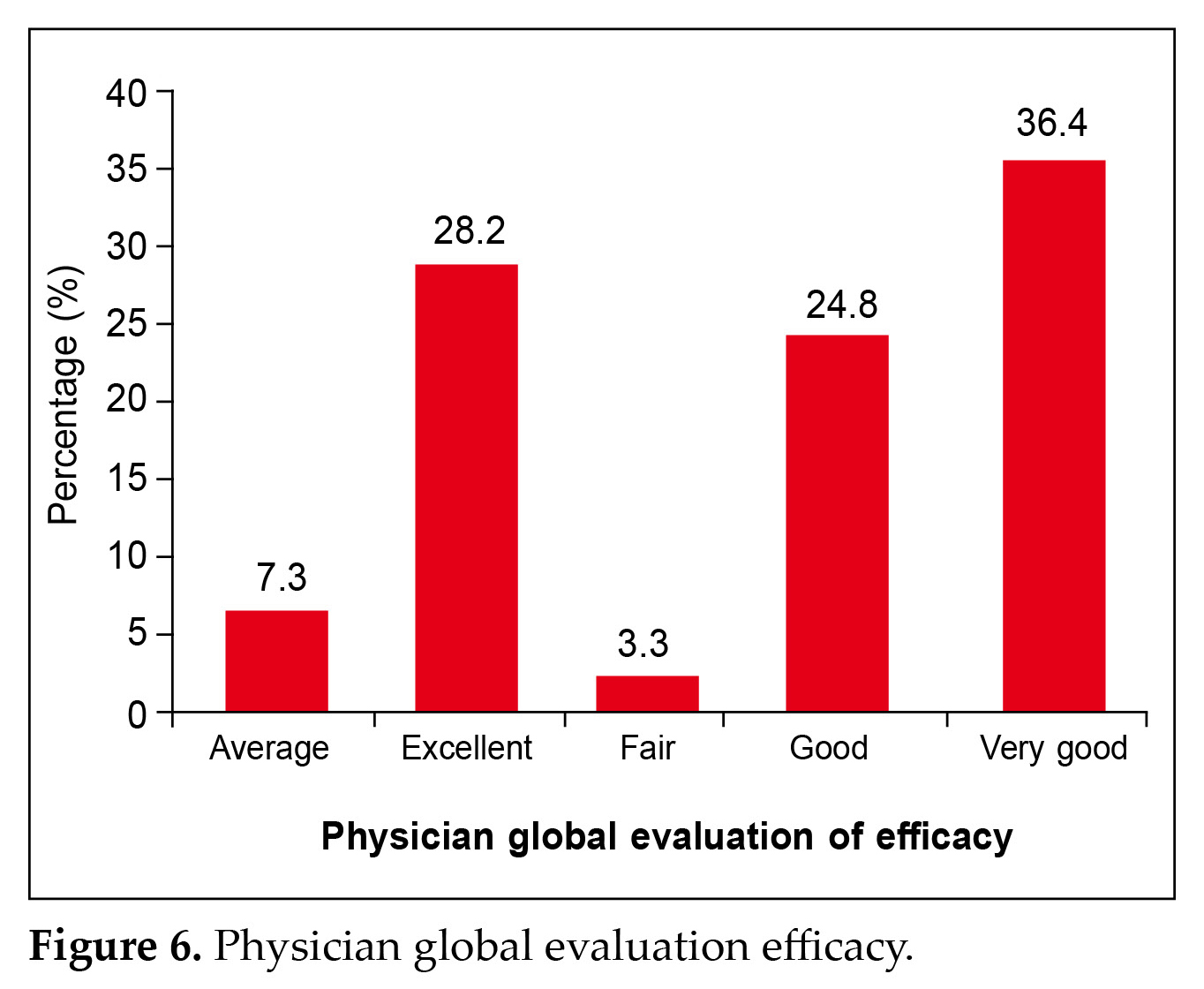
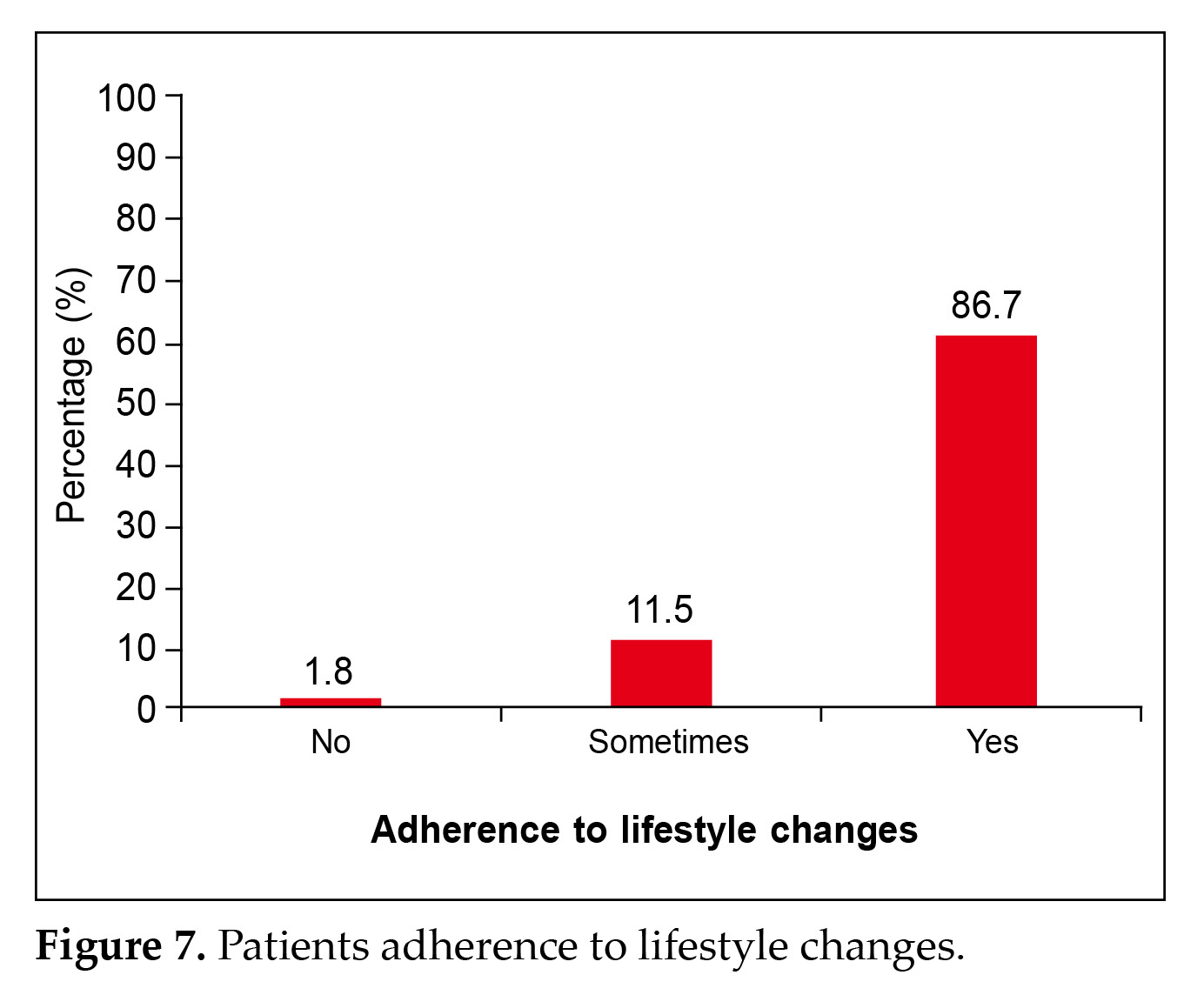
It was further observed that 36.4% physician had a view that the switching was having very good efficacy, followed by 28.2% physician having a view of excellent efficacy as is evident from Figure 6; 86.7% patients adhered to proper lifestyle changes as seen in Figure 7.
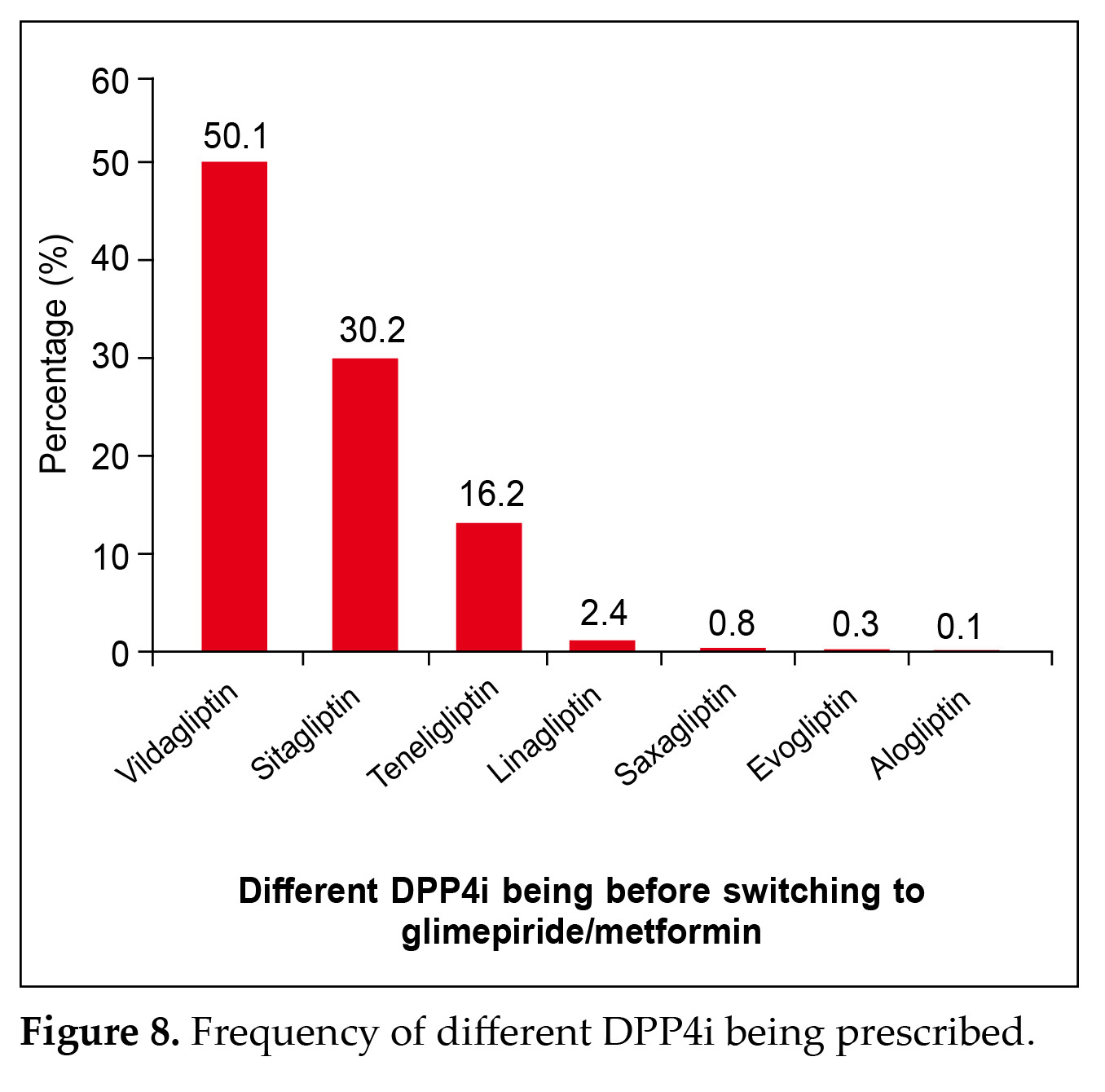
The survey also gave a clear picture of the DPP4i which was being prescribed the most along with metformin to the patient before switching to glimepiride/metformin. Vildagliptin (50%) was the most common DPP4i being prescripbed, followed by sitagliptin (30.2%), teneligliptin (16.0%), linagliptin (2.4%), saxagliptin (0.8%), evogliptin (0.3%) and alogliptin (0.1%) as is observed from Figure 8.
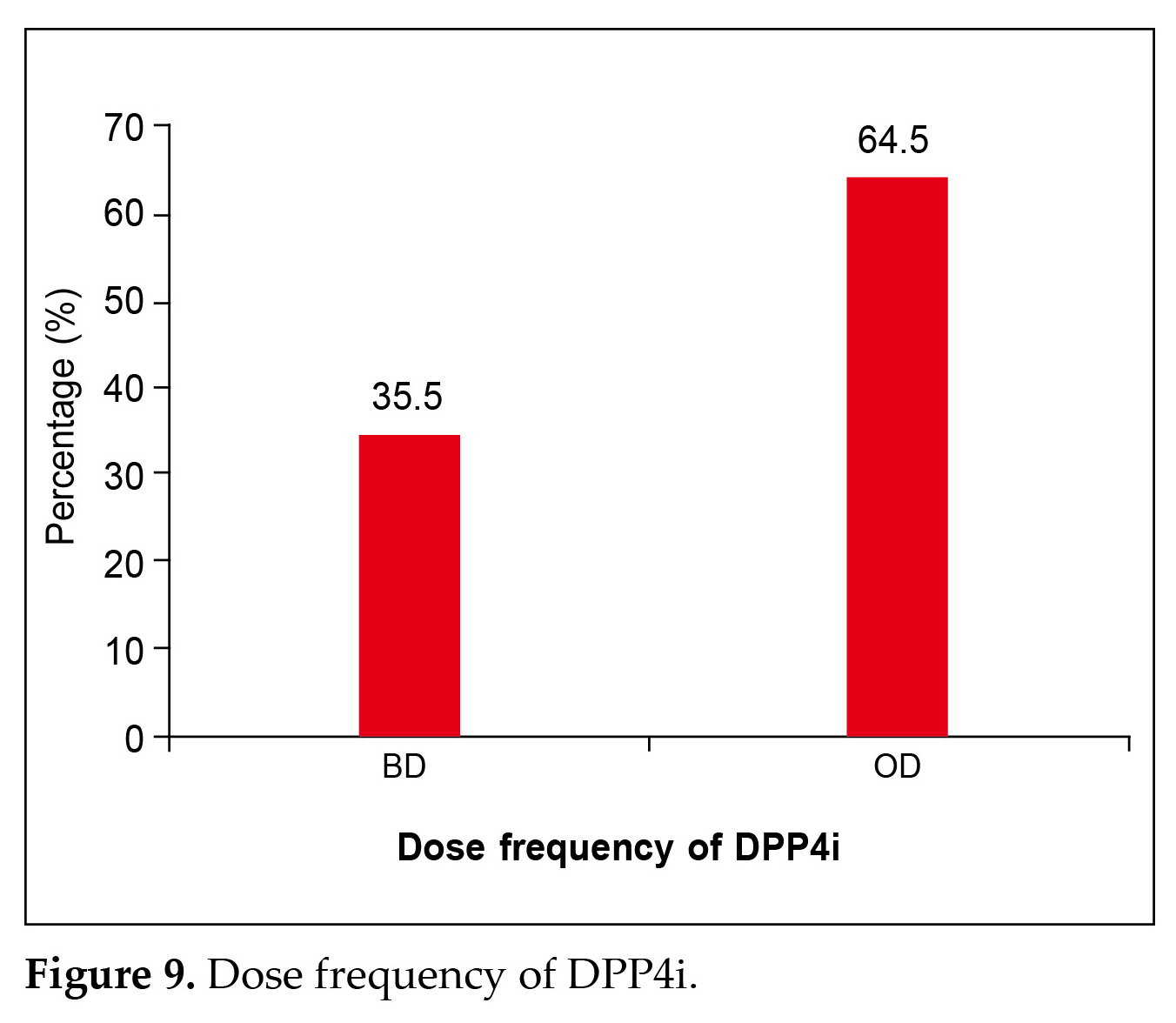
Further OD (once a day) dose was the most prevalent (64.5%) dose of DPP4i being prescribed by the physicians, followed by BD (twice a day) with 35.5% as seen on Figure 9.
The median dose of DPP4i used was found to be 50 with interquartile range (IQR) of 30. More specifically, the median dose of glimepiride used was 1 with IQR of (1.5) and the median dose of metformin used was 500 with IQR of 350. It was further observed
that DPP4i being prescribed in patients as add-on to metformin was highest (69.2%), followed by first-line combination therapy with metformin (30.5%) as seen in Figure 10. The most important reason for switching to glimepiride/metformin was found to improve HbA1c (71.7%), followed by aiming to control the uncontrolled

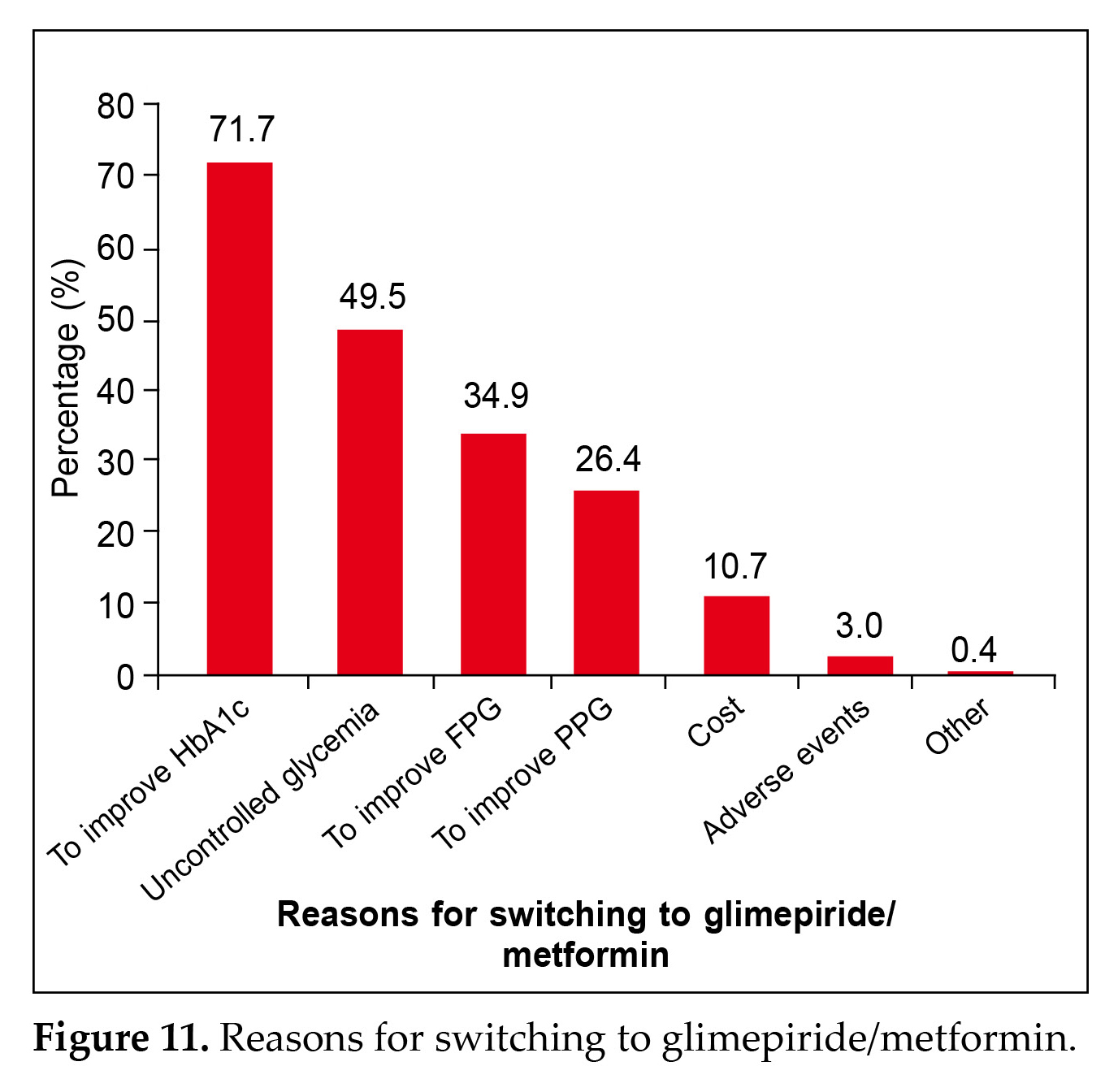
glycemia accounting for 49.5% of patients, to improve FPG (34.9%), to improve PPG (26.4%), cost-effectiveness (10.7%), less adverse effects (3%), and other factors accounted for only 0.4% as is evident from Figure 11.
Discussion
Dipeptidyl-peptidase-4 inhibitors (DPP4i), in general, are recommended as second- and third-line therapy for T2DM and offer the option for improvement in both HbA1c and beta-cell survival, but a long-term clinical trial data are not yet available to assess the sustainability of glycemic control and protection of beta-cell mass10.
Some reasons for switch from DPP4i to modern SUs might include modest glycemic lowering, relatively higher cost and reported incidences of pancreatic disease, arthritis, bullous pemphigoid (BP) some cases of heart failure11. Detailed effects of the drug combination on different parameters such as fasting blood glucose (FBG), PPG, HbA1c, and body weight are included. It further provides the clinical evidence on switching to modern SU/metformin in patients uncontrolled on DPP4i-based therapies.
The study included, 2,736 T2DM patients who were 18 years and above with an average age of 38.46 ± 7.21. It was observed that switching from DPP4i to modern SU/metformin combination was beneficial for patients who could not control their hyperglycemia even with DPP4i.
HbA1c reduced from 9.64 ± 1.79 before treatment to 7.52 ± 1.97 after treatment. Similarly, FPG which was 175.14 ± 89.89 mg/dL reduced to 133.37 ± 43.59 mg/dL after treatment. PPG lowered from 251.38 ± 80.30 mg/dL before treatment to 183.98 ± 54.76 mg/dL after treatment. Being overweight or obese significantly raises the risk of developing diabetes. Research indicates that approximately 86% of adults with T2D fall into the overweight or obese category12,13.
A similar trend was also observed in the current study. About 28.1% were obese and 49.2% of the participants were overweight.
Studies have shown that the majority of the 463 million individuals who have diabetes globally reside in LMICs (low- and middle-income countries). Additionally, it was shown that less than 1 in 10 diabetics in LMICs are treated with complete, guideline-based care14.
The Centers for Disease Control and Prevention (CDC) advises that engaging in physical activity not only helps regulate blood sugar levels but also reduces the risk of heart disease and nerve damage. They recommend 150 minutes of moderate-intensity physical activity per week15.
In the present study, the demographic details showed that 61.5% of the participants belonged to the economically weaker section. About 45.5% were moderately active, while 26.4% were engaged in regular exercise and 13.2% were inactive as shown in Figure 2.
Research has established a mutual relationship between COVID-19 and diabetes mellitus. Diabetic individuals infected with severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) face an increased risk of hospitalization, severe pneumonia, and mortality compared to those without diabetes. The key characteristics of diabetes, namely insulin deficiency and elevated blood glucose levels, are exacerbated by SARS-CoV-2’s ability to target and harm the body’s insulin-producing cells16,17. In the present study, it was seen that 25.1% of the participating population had a history of COVID-19 as illustrated in Figure 3.
DPP4i typically do not lead to weight gain and carry a low risk of hypoglycemia and other side effects. While they generally have a minimal impact on weight, some patients with lower baseline blood sugar levels have experienced slight weight reduction when using the DPP4i vildagliptin. This weight-neutral effect of vildagliptin may be attributed, at least in part, to its inherently low risk of hypoglycemia18,19.
A similar observation was also seen in this study with 94.8% of patients reporting no hypoglycemic events (Fig. 4) and 96.2% did not gain weight after switching to glimepiride/metformin (Fig. 5).
The combination of glimepiride/metformin achieves good glycemic control and tolerability. In a recent study, Kumar also reported a similar finding that stated the efficacy and tolerability to be good to excellent (97.3% and 96.6%) in a vast majority of patients20.
In another international prospective study, diabetic patients treated with glimepiride showed fewer hypoglycemic episodes compared to those treated with glibenclamide. Glimepiride’s documented cardiovascular safety/neutrality and reduced hypoglycemia episodes make it an attractive alternative for the management of persons with long-standing diabetes21,22.
Modern SUs (glimepiride/glibenclamide, etc.) offer superior glycemic efficacy, has better cardiovascular profile and are also available at a reasonable cost. Treatment with modern SUs is associated with a lower economic burden, and hence they are an effective alternative to other newer antidiabetic drugs23,24.
It was further observed that 36.4% physician had a view that the switching was having very good efficacy, followed by 28.2% physician having a view of excellent efficacy as is evident from Figure 6. This is in line with a recent retrospective, nonrandomized, noncomparative, multicentric real-world study which showed that glimepiride and metformin combinations are frequently prescribed in diabetes with comorbidities like hypertension and dyslipidemia and complications for the best glycemic control25.
Most of the global bodies and guidelines advise metformin and changes in the lifestyle for treating newly diagnosed T2DM, with variations mainly in the second- and third-line antidiabetic agents26,27. It was seen that 86.7% of the participating patients adhered to proper lifestyle changes as seen in Figure 7. The study also found that vildagliptin (50%) was the most common DPP4i being prescribed, along with metformin to the patient before switching to glimepiride/metformin.
It was followed by sitagliptin (30.2%) as is observed from Figure 8 below.
Vildagliptin is a potent and selective inhibitor of DPP-4.
It enhances glycemic control by increasing the responsiveness of both islet alpha-cells and beta-cells to glucose. When used in combination with metformin, pioglitazone, glimepiride, or insulin, vildagliptin leads to significant additional reductions in HbA1c levels in patients. Moreover, it has been found to reduce the occurrence of hypoglycemic episodes when added to insulin therapy. Preliminary findings suggest that the enhanced function of islet cells, which contributes to the effectiveness of vildagliptin in treating T2D, is also evident in individuals with impaired glucose tolerance. Vildagliptin treatment in such cases results in decreased fluctuations in blood sugar levels28.
Besides being an antidiabetic drug vildagliptin also possesses a number of other pharmacological features, such as neuroprotective benefits in vivo and in vitro models29.
Vildagliptin also causes a dosage-related reduction in HbA1c and FPG when added to a steady dose of metformin. Furthermore, metformin increases vildagliptin’s ability to boost plasma levels of intact glucagon-like peptide-1 (GLP-1), which is one of the main ways that vildagliptin’s therapeutic effects are mediated via
GLP-130. The above factors make vildagliptin a good candidate to top the list of DPP4i, which are generally prescribed along with glimepiride/metformin combination.
Vildagliptin and metformin were observed to significantly lower plasma glucose and HbA1c when taken once daily, suggesting that this regimen may be a more practical and affordable beginning point for treatment than a twice-daily regimen31.
Hence, in the current study also OD (once a day) dose was the most prevalent (64.5%) dose of DPP4i being prescribed by the physicians, followed by BD (twice a day) with 35.5% as seen on Figure 9. The median dose of DPP4i used was found to be 50 with IQR of 30.
More specifically, the median dose of glimepiride used was 1 with IQR of (1.5) and the median dose of metformin used was 500 with IQR of 350. Vildagliptin, when used as add-on therapy to metformin, improved Chinese patients’ glycemic control and was well-tolerated32. Further, a study found that individuals with T2DM who had poor glycemic control benefited from adding vildagliptin to their regimen of metformin and glimepiride33. A similar observation was seen in the present study.
DPP4i was being prescribed in patients as add-on to metformin was highest (69.2%), followed by first-line combination therapy with metformin (30.5%) as seen in Figure 10.
In the current study, a significant decrease in the FBG, PPG, and HbA1c was observed, which is in similar lines with the findings by Phung et al (2010), Hassan and Abd-Allah (2015), Kumar (2021), Shrivastava et al (2023)20,34-36.
The most important reason for switching to glimepiride/metformin was found to improve HbA1c (71.7%), followed by aiming to control the uncontrolled glycemia accounting for 49.5% of patients, to improve FPG (34.9%), to improve PPG (26.4%), cost-effectiveness (10.7%), less adverse effects (3%), and other factors accounted for only 0.4% as is evident from Figure 11.
Numerous studies have demonstrated that incorporating glimepiride into the treatment regimen of T2D patients who were not achieving adequate glycemic control with metformin alone led to improved blood sugar management. Furthermore, the concurrent administration of glimepiride and metformin in a single medication form proved to be both effective and safe for individuals with T2D37-39..
A study found that in T2D patients whose condition was not properly managed by low-dose metformin monotherapy, glimepiride/metformin fixed-dose combination treatment was more successful in glucose control than metformin uptitration and was well-tolerated40.
This study also reported that only 5.2% of patients
(p < 0.001) experienced hypoglycemia after switching to glimepiride/metformin combination. The combination of glimepiride/metformin achieves good glycemic control and tolerability. In a recent investigation, Prasanna Kumar et al similarly reported findings indicating that the majority of patients experienced a high level of effectiveness and tolerability, with rates reaching 97.3% and 96.6%, respectively41.
In another worldwide prospective research it was found that diabetic individuals on glimepiride experienced fewer hypoglycemia episodes than those taking glibenclamide42. Glimepiride is a desirable choice for the management of people with long-term diabetes due to its shown cardiovascular safety/neutrality and decreased hypoglycemic episodes43.Modern SUs have better cardiovascular profiles, greater glycemic effectiveness, and are also reasonably priced. Modern SUs are an efficient alternative to other more recent antidiabetic medications since they are connected with a smaller financial burden during treatment23. According to a research by Barnett et al
(2015), metformin plus SU, thiazolidinedione, or sodium glucose co-transporter 2 (SGLT2) inhibitor medication were typically well-tolerated and improved glycemic parameters when combined with a DPP4i44.
Decisions regarding treatment are determined by considering factors such as the effectiveness of glycemic control, safety profiles, and the impact of the therapy on weight and the risk of hypoglycemia, existing comorbidities, and treatment costs. Switching to modern SU/metformin in patients uncontrolled on DPP4i-based therapies was an beneficial alternative for diabetes management.
Limitations
As the study was a multicenter observational survey it had limitations such as selection and response bias. Diverse patient populations across centers made the generalization difficult.
Conclusion
The retrospective clinical evidence has shed light on the clinical outcomes associated with transitioning patients from DPP4i-based therapies to modern SU/metformin combinations.
The study found that the transition to modern SU/metformin combinations in DPP4i-uncontrolled patients carries significant implications for diabetes management. It could play a crucial role in informing treatment paradigms. The transition provides an alternative treatment option in addressing inadequate glycemic control with DPP4i-based therapies and thus improves diabetes care, enhancing the quality of life for patients facing the challenges of diabetes management.
Major Findings
Switching to modern SU/metformin showed significant improvement in the HbA1c values and FPG and PPG levels and were controlled well.
Majority (94.8%) of patients had no hypoglycemic events and 96.2% did not gain weight after switching to SU/metformin.
Vildagliptin (50%) was the most common DPP4i being prescribed, followed by sitagliptin (30.2%).
The main reason of switching to SU/metformin was to improve the HbA1c levels, followed by controlling the uncontrolled glycemic levels and further improving FPG and PPG levels.
Transitioning to SU and metformin combo is an effective choice for diabetes when DPP4i fall short.
References
- Wood S, Magliano DJ, Bell JS, Shaw JE, Ilomäki J. Treatment dynamics in people who initiate metformin or sulfonylureas for type 2 diabetes: a national cohort study. Front Pharmacol. 2021;12:794273.
- Pantalone KM, Misra-Hebert AD, Hobbs TM, Ji X, Kong SX, Milinovich A, et al. Clinical inertia in type 2 diabetes management: evidence from a large, real-world data set. Diabetes Care. 2018;41(7):e113-4.
- Committee ADAPP. 13. Older Adults: Standards of Care in Diabetes—2024. Diabetes care. 2023;47(Supplement_1):S244-S57.
- Huo L, Magliano DJ, Rancière F, Harding JL, Nanayakkara N, Shaw JE, et al. Impact of age at diagnosis and duration of type 2 diabetes on mortality in Australia 1997-2011. Diabetologia. 2018;61(5):1055-63.
- Yazbeck R, Jaenisch SE, Abbott CA. Dipeptidyl peptidase 4 inhibitors: applications in innate immunity? Biochem Pharmacol. 2021;188:114517.
- Daza-Arnedo R, Rico-Fontalvo JE, Pájaro-Galvis N, Leal-Martínez V, Abuabara-Franco E, Raad-Sarabia M, et al. Dipeptidyl peptidase-4 inhibitors and diabetic kidney disease: a narrative review. Kidney Med. 2021;3(6):1065-73.
- Rehman MB, Tudrej BV, Soustre J, Buisson M, Archambault P, Pouchain D, et al. Efficacy and safety of DPP-4 inhibitors in patients with type 2 diabetes: meta-analysis of placebo-controlled randomized clinical trials. Diabetes Metab. 2017;43(1):48-58.
- Lee SG, Lee HJ, Yoon MS, Kim DH. Association of dipeptidyl peptidase 4 inhibitor use with risk of bullous pemphigoid in patients with diabetes. JAMA Dermatol. 2019;155(2):172-7.
- Fernandes G, Matos JE, Jaffe DH, Beyer G, Yang L, Iglay K, et al. Factors associated with the discontinuation of dipeptidyl peptidase-4 inhibitors (DPP-4is) after initiation of insulin. Curr Med Res Opin. 2020;36(3):377-86.
- Brock CM. DPP-4 inhibitors: What is their place in therapy? US Pharm. 2010;35(5)(Diabetes suppl):8-13.
- Feingold KR, Oral and Injectable (Non-Insulin) Pharmacological Agents for the Treatment of Type 2 Diabetes. [Updated 2022 Aug 26]. 2022 [cited 2023 20-07-23]. Available from: https://www.semanticscholar.org/paper/Oral-and-Injectable-(Non-Insulin)-Pharmacological-2-Feingold/7b3c907a51e314fb3d09da78a0575a95694530d9.
- Aras M, Tchang BG, Pape J. Obesity and Diabetes. Nurs Clin North Am. 2021;56(4):527-41.
- Overweight and obesity in people with type 1 diabetes nearly same as general population. Published in February 13, 2023. Available from: https://publichealth.jhu.edu/2023/overweight-and-obesity-in-people-with-type-1-diabetes-nearly-same-as-general-population. Accessed September 86, 2024.
- Flood D, Seiglie JA, Dunn M, Tschida S, Theilmann M, Marcus ME, et al. The state of diabetes treatment coverage in 55 low-income and middle-income countries: a cross-sectional study of nationally representative, individual-level data in 680 102 adults. Lancet Healthy Longev. 2021;2(6):e340-51.
- Get Active! Available from: https://www.cdc.gov/diabetes/managing/active.html. Accessed October 9, 2024.
- Lima-Martínez MM, Carrera Boada C, Madera-Silva MD, Marín W, Contreras M. COVID-19 and diabetes: a bidirectional relationship. Clin Investig Arterioscler. 2021;33(3):151-7. English, Spanish.
- How COVID-19 can lead to diabetes. 2021. Available from: https://www.nih.gov/how-covid-19-can-lead-diabetes. Accessed September 6, 2024.
- Ahrén B. DPP-4 inhibition and the path to clinical proof. Front Endocrinol (Lausanne). 2019;10:376.
- Foley JE, Jordan J. Weight neutrality with the DPP-4 inhibitor, vildagliptin: mechanistic basis and clinical experience. Vasc Health Risk Manag. 2010;6:541-8.
- Kumar S. Comparison of safety and efficacy of glimepiride-metformin and vildagliptin-metformin treatment in newly diagnosed type 2 diabetic patients. Indian J Endocrinol Metab. 2021;25(4):326-31.
- Das AK, Saboo B, Chawla R, Aravind SR, Rajput R, Singh AK, et al. Time to reposition sulfonylureas in type 2 diabetes management in Indian context: a pragmatic practical approach. Int J Diabetes Dev Ctries. 2023:1-19.
- Kalra S, Das AK, Baruah MP, Unnikrishnan AG, Dasgupta A, Shah P, et al. Glucocrinology of modern sulfonylureas: clinical evidence and practice-based opinion from an International Expert Group. Diabetes Ther. 2019;10(5):1577-93.
- Kalra S, Ghosh S, Das AK, Nair T, Bajaj S, Priya G, et al. Unravelling the utility of modern sulfonylureas from cardiovascular outcome trials and landmark trials: expert opinion from an international panel. Indian Heart J. 2020;72(1):7-13.
- Sahay RK, Mittal V, Gopal GR, Kota S, Goyal G, Abhyankar M, et al. Glimepiride and metformin combinations in diabetes comorbidities and complications: real-world evidence. Cureus. 2020;12(9):e10700.
- Hassanein M, Al Sifri S, Shaikh S, Abbas Raza S, Akram J, Pranoto A, et al; DIA-RAMADAN study investigators. A real-world study in patients with type 2 diabetes mellitus treated with gliclazide modified-release during fasting: DIA-RAMADAN. Diabetes Res Clin Pract. 2020;163:108154.
- World Health Orgamization. Continuity of essential health services: facility assessment tool: a module from the suite of health service capacity assessments in the context of the COVID-19 pandemic: interim guidance 20 October 2020. Geneva: World Health Organization; 2020.
- Type 2 diabetes in adults: management. NICE guideline [NG28]: NICE. Published in 2015. Last updated June 29, 2022. Available from: https://www.nice.org.uk/guidance/ng28. Accessed September 10, 2024.
- Panina G. The DPP-4 inhibitor vildagliptin: robust glycaemic control in type 2 diabetes and beyond. Diabetes Obes Metab. 2007;9 Suppl 1:32-9.
- Pariyar R, Bastola T, Lee DH, Seo J. Neuroprotective effects of the DPP4 inhibitor vildagliptin in in vivo and in vitro models of Parkinson's disease. Int J Mol Sci. 2022;23(4):2388.
- Ahrén B, Foley JE, Bosi E. Clinical evidence and mechanistic basis for vildagliptin's action when added to metformin. Diabetes Obes Metab. 2011;13(3):193-203.
- Chatterjee S, Chatterjee S. Vildagliptin with metformin once-daily regimen-insights from a single-center analysis. Am J Ther. 2015;22(3):195-8.
- Pan C, Xing X, Han P, Zheng S, Ma J, Liu J, et al. Efficacy and tolerability of vildagliptin as add-on therapy to metformin in Chinese patients with type 2 diabetes mellitus. Diabetes Obes Metab. 2012;14(8):737-44.
- Lukashevich V, Del Prato S, Araga M, Kothny W. Efficacy and safety of vildagliptin in patients with type 2 diabetes mellitus inadequately controlled with dual combination of metformin and sulphonylurea. Diabetes Obesity Metab. 2014;16(5):403-9.
- Phung OJ, Scholle JM, Talwar M, Coleman CI. Effect of noninsulin antidiabetic drugs added to metformin therapy on glycemic control, weight gain, and hypoglycemia in type 2 diabetes. JAMA. 2010;303(14):1410-8.
- Hassan MH, Abd-Allah GM. Effects of metformin plus gliclazide versus metformin plus glimepiride on cardiovascular risk factors in patients with type 2 diabetes mellitus. Pak J Pharm Sci. 2015;28(5):1723-30.
- Shrivastava A, Kesavadev J, Mohan V, Saboo B, Shrestha D,
Maheshwari A, et al. Clinical evidence and practice-based guidelines on the utility of basal insulin combined oral therapy (metformin and glimepiride) in the current era. Curr Diab Rev. 2023;19(8):e090123212444.
- Kalra S, Bahendeka S, Sahay R, Ghosh S, Md F, Orabi A, et al. Consensus recommendations on sulfonylurea and sulfonylurea combinations in the management of type 2 diabetes mellitus - International Task Force. Indian J Endocrinol Metab. 2018;22(1):132-57.
- Charpentier G, Fleury F, Kabir M, Vaur L, Halimi S. Improved glycaemic control by addition of glimepiride to metformin monotherapy in type 2 diabetic patients. Diabet Med. 2001;18(10):828-34.
- González-Ortiz M, Martínez-Abundis E; Grupo para el Tratamiento de la Diabetes Mellitus con Combinaciones. Efficacy and safety of glimepiride plus metformin in a single presentation, as combined therapy, in patients with type 2 diabetes mellitus and secondary failure to glibenclamide, as monotherapy. Rev Invest Clin. 2004;56(3):327-33. Spanish.
- Kim HS, Kim DM, Cha BS, Park TS, Kim KA, Kim DL, et al. Efficacy of glimepiride/metformin fixed-dose combination vs metformin uptitration in type 2 diabetic patients inadequately controlled on low-dose metformin monotherapy: a randomized, open label, parallel group, multicenter study in Korea. J Diabetes Investig. 2014;5(6):701-8.
- Prasanna Kumar KM, Seshadri K, Aravind SR, Deb P, Modi KD, Gopal RA, et al. Real-world observational study of glimepiride and metformin fixed-dose combination along with insulin in the management of type 2 diabetes mellitus: Indian experience. Cureus. 2021;13(1):e13020.
- Zekry R, Omran GA, El-Gharbawy NM, Werida RH. Comparative study of Dapagliflozin versus Glimepiride effect on insulin regulated aminopeptidase (IRAP) and interleukin-34 (IL-34) in patient with type 2 diabetes mellitus. Sci Rep. 2023;13(1):6302.
- ElSayed NA, Aleppo G, Aroda VR, Bannuru RR, Brown FM,
Bruemmer D, et al; American Diabetes Association. 9. Pharmacologic Approaches to Glycemic Treatment: Standards of Care in Diabetes-2023. Diabetes Care. 2023;46(Suppl 1):S140-57.
- Barnett AH, Charbonnel B, Moses RG, Kalra S. Dipeptidyl peptidase-4 inhibitors in triple oral therapy regimens in patients with type 2 diabetes mellitus. Curr Med Res Opin. 2015;31(10):1919-31.