Abstract
Diabetes presents a significant public health challenge in Nepal, compounded by its diverse topography and cultural beliefs. Despite a historical emphasis on patient-centered care, which fosters unity among physician, patient, drug, and caretaker, effective diabetes management is hindered by lack of awareness, cultural preferences for alternative therapies, and limited
health care resources. Addressing these challenges requires a consensus to optimize insulin's historical significance, marking a century as the first antidiabetic agent. Although advancements have been made, a gap persists in understanding, awareness, and utilization of insulin among Nepalese physicians, necessitating tailored management approaches. A consensus meeting of leading experts and diverse advisors from Nepal highlights the need for collective guidelines to optimize insulin usage. Nepalese patients often exhibit reluctance towards insulin therapy, stemming from concerns about its complexity and efficacy. Blood glucose monitoring is vital for guiding insulin regimens, especially in critically ill patients, with the SECURE model offering a comprehensive management approach. Tailoring insulin regimens to individual lifestyles enhances treatment adherence and overall efficacy. A patient-centered approach is paramount in optimizing diabetes management in Nepal. Through collective agreement and guidelines, health care professionals can improve their knowledge and confidence in insulin therapy, leading to better patient outcomes and public health.
Keywords: Type 2 diabetes mellitus, insulin, patient-centered, Nepal
Introduction
Diabetes is recognized as a serious public health concern with a considerable impact on human life and health expenditures. Rapid economic development and urbanization have led to a rising burden of diabetes in many parts of the world. According to the 2021 report by the International Diabetes Federation (IDF), a staggering 537 million adults globally were affected by diabetes, with 79.4% of cases concentrated in these lower-income countries, impacting individuals aged 20 to 79 years1. In Southeast Asia, the prevalence of diabetes was 8.8% in 2019, and it’s expected to increase to 9.7% by 20302.
In Nepal, diabetes ranks as the third most common noncommunicable disease, with a prevalence ranging from 6.3% to 25.9%3. The country's diverse topography, spanning from the fertile Gangetic plains to the frozen Himalayan mountains, presents unique challenges in addressing diabetes. Due to this diversity, tailored approaches are required for different regions4. Additionally, the prevalence of type 2 diabetes mellitus (T2DM), prediabetes and impaired glucose tolerance stands at 10%, 19.4%, and 11% respectively5.
Cases of type 1 diabetes mellitus (T1DM) in Nepal have increased significantly from 154.53 cases per 100,000 population in 1990 to 201.99 cases per 100,000 population in 20196. The country’s geographical diversity results in a wide range of climates, from scorching temperatures exceeding 45°C in the Tarai plains to alpine climates with temperatures below -30°C in the northern Himalayan region. Insulin emerges as the cornerstone of medical management for T1DM and is also utilized in individuals with uncontrolled T2DM, even when optimal oral antidiabetic drugs are being used. Customized strategies are necessary to navigate the challenges of diabetes across these varied regions and meet the specific needs of each area4.
The concept of patient-centered care, as emphasized in recent diabetes guidelines, resonates deeply with ancient practices rooted in Indian history. While the term itself may be modern, the essence of patient-centered care can be traced back centuries. The famous Ayurvedic physician Atreya described the quadruple, which suggests that the patient is an integral part of the four equally important "angles" required for effective treatment. The other three angles mentioned by Atreya are the physician, the drug, and the attendant7. The quadruple concept of Atreya is culturally relevant to Nepal which emphasizes the necessity of unity among physician, patient, drug, and caretaker. Person-centered care prioritizes the individual with the disease, necessitating effective communication for cohesive teamwork. In Nepal, health care providers must ensure clear information for individuals with diabetes, empowering them in health care decisions. This approach improves outcomes and acknowledges unique circumstances. Beyond medical and nursing personnel, responsibility extends to family, policymakers, and civil society, forming a comprehensive strategy for managing diabetes8,9. Addressing the challenge of diabetes management in Nepal is undeniably complex due to a range of contributing factors.
Need for Consensus
The need for a consensus is to optimize the historical significance of insulin as the first antidiabetic agent available for 100 years. From its initial poorly defined extracts of animal pancreatic origin, insulin has evolved into pure and precisely controlled formulations designed to mimic physiological insulin release patterns. Despite these advancements, there remains a considerable grey area concerning the understanding, awareness, and utilization of insulin among practicing physicians in Nepal. Also, looking at the diverse lifestyles and landscapes of Nepal, there is a need for tailored approaches to the management of diabetes in different regions. To optimize diabetes management and ensure better patient outcomes, a collective agreement and guidelines on the appropriate use of insulin are imperative. This consensus will foster improved knowledge and confidence among health care professionals, leading to more effective and patient-centric insulin therapy in Nepal.
Methodology
A group of endocrinologists and diabetes specialists in Nepal held a focused board meeting to discuss the person-centered insulin approach for the management of diabetes mellitus. The meeting was moderated by leading endocrinologists from India and a panel of advisors across Nepal.
Role and Importance of Insulin in the Management of Diabetes Mellitus
The lack of awareness and understanding about diabetes within the community, along with a reluctance to adopt medication or advance treatment, pose significant hurdles. Additionally, preference for alternative therapies and limited health care resources exacerbate these challenges. The inadequate availability of diagnostic and laboratory facilities compounds these issues. Cultural beliefs surrounding treatment greatly influence patients' adherence to prescribed regimens, often deeply rooted in Nepalese society's sociocultural context. Limited exploration of specific sociocultural traditions and beliefs related to diabetes treatment further complicates matters10. Research reveals that Nepalese patients perceive antidiabetic medications as potentially harmful and irreversible, impacting their willingness to initiate treatment and compromising adherence11. To effectively address patients' needs, a fundamental shift in the approach to diabetes management and attitudes toward insulin use is crucial. Reluctance to incorporate insulin therapy stems from perceived complexity, doubts about efficacy, and concerns regarding hypoglycemic episodes and weight gain12.
Insulin therapy is vital for managing diabetes, particularly in T1DM, where lifelong replacement therapy is essential. Treatment options include multiple daily injections or continuous subcutaneous insulin infusion13.
For newly diagnosed T2DM patients, initial treatment often involves dietary adjustments, exercise, and oral medications, but insulin may be necessary if blood sugar levels remain uncontrolled14.
The American Association of Clinical Endocrinologists recommends considering insulin when A1c levels are >9% or when oral therapy fails. Insulin helps restore beta cells and can be used alone or with oral medications, especially for hospitalized or critically ill patients and those with end-stage liver disease or liver failure15. Insulin glargine is commonly prescribed and helps preserve beta-cell function when initiated early16. (Table 1 and 2).
Benefits of Early Initiation and Intensification
Insulin is clearly the most effective way to control blood glucose.
Better glycemic control to reduce the incidence and severity of long-term vascular outcomes.
Early insulin supplementation may alter the progressive course of diabetes.
Restores the function of beta cells and significantly reduces insulin resistance.
Early initiation of insulin therapy improves beta-cell function and mass by inducing ‘beta-cell rest’.
When to Initiate Insulin?
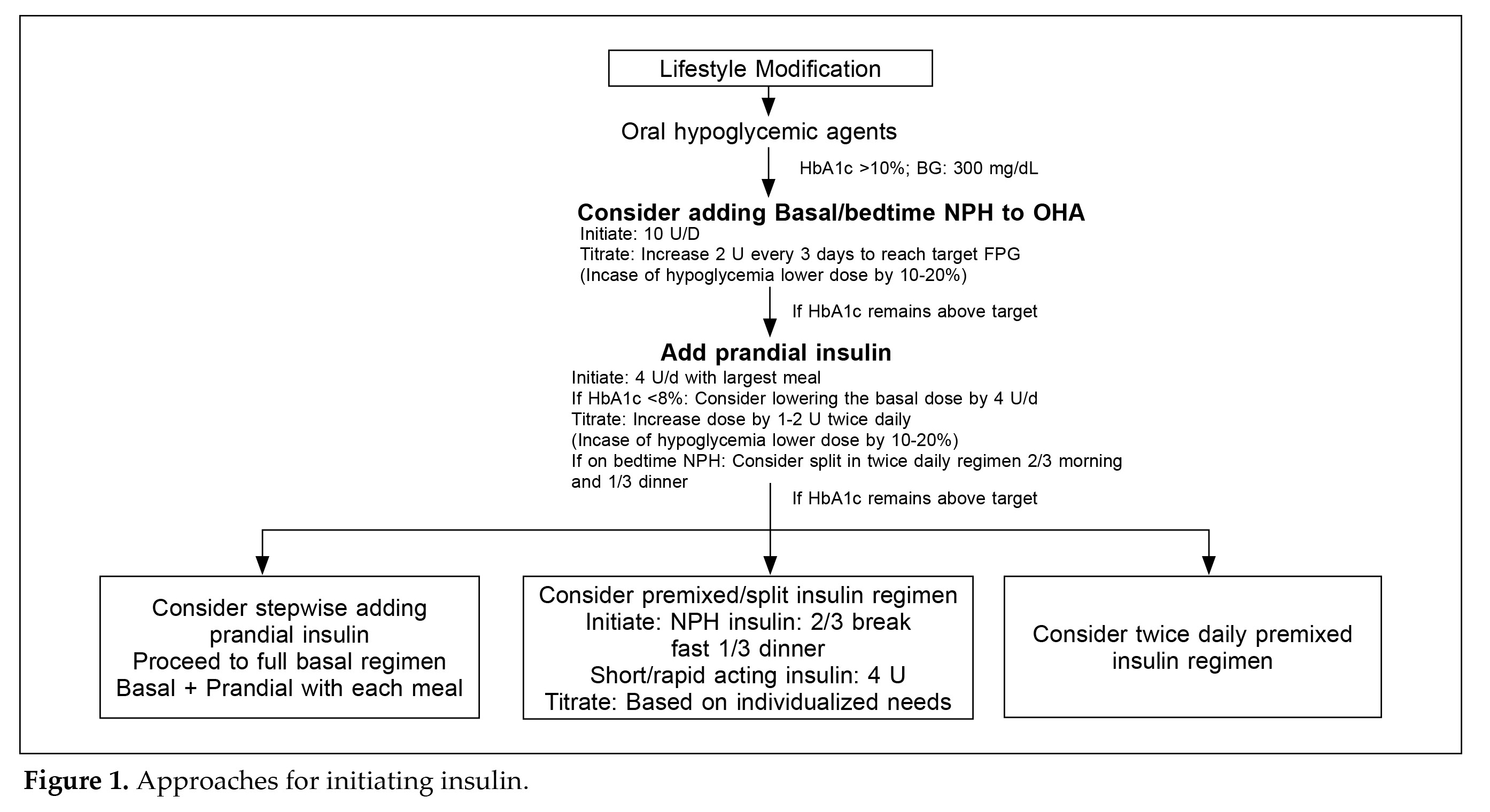
In managing diabetes, glycemic treatment should follow a stepwise approach, swiftly introducing successive interventions after treatment failure (A1c ≥10%). Initiation of insulin is recommended when A1c reaches ≥10% after 2-3 months of dual oral therapy, with once-daily basal insulin being the preferred regimen. Timely initiation and rapid titration of the insulin dose are crucial for successful therapy, and the risk of hypoglycemia is low in patients starting insulin therapy, making Neutral Protamine Hagedorn (NPH) insulin the most cost-effective option. If glycemic goals are still not achieved despite successful basal insulin titration (fasting plasma glucose ≤100 mg/dL) or if titration is
|
Table 1. Types of Insulin Available in Nepal
|
|
Long acting
|
Intermediate acting
|
Short acting
|
Rapid acting
|
Premixed insulin
|
|
Human insulin
|
Analogues
|
|
Insulin
Glargine
|
|
|
Insulin
Lispro
|
30/70
Regular/NPH
|
Insulin Degludec + Insulin Aspart
|
|
Insulin detemir
|
|
|
Insulin
Glulisine
|
50/50
Regular/NPH
|
Insulin Aspart Biphasic
|
| |
Insulin
(Human) NPH
|
Insulin
(Human)
Regular
|
|
|
30/70 Biphasic Aspart/Aspart protamine
|
|
Insulin
Degludec
|
|
|
Insulin
Aspart
|
|
50/50 Biphasic Aspart/Aspart protamine
|
| |
|
|
|
|
25/75 Biphasic Lispro/Lispro protamine
|
| |
|
|
|
|
50/50 Biphasic Lispro/Lispro protamine
|
NPH: Neutral protamine hagedorn
|
Table 2. List of Available Insulin Delivery Devices and Glucose Monitoring tools in Nepal
|
|
Insulin pen
|
Glucose monitoring tools
|
|
Lupisulin pen
Basugine Pen
Humalog pen
Levemir Flexpen
Ryzodeg pen
Novomix pen
NovoRapid pen
Sanofi Allstar
|
POC assessment tools like one touch/Morepen’s
On call plus
Accu-Chek
VivaChek
Clever chek Sinocare
Gluco One
|
POC: Point-of-care
limited by hypoglycemia, treatment intensification is needed, involving the addition of prandial or biphasic insulin to the regimen17. (Fig 1)
Ethnic Differences in Nepalese Patients are Based on Diet and Lifestyle
The sociocultural traditions and beliefs of Nepal and the Nepalese population have not been extensively explored in relation to diabetes treatment10.
Diet Pattern
In Nepal, the traditional eating patterns involve two main meals at around 9 am and 6 pm, accompanied by small snacks and tea in between. However, Nepal is witnessing dietary changes akin to the high-fat, high-sugar, and high-meat consumption patterns prevalent in Western countries. The consumption of refined grains, meat, and alcohol is linked to a higher prevalence of overweight, while fast food intake is associated with a higher prevalence of obesity in older adults (40 years and above)18.
Contribution of FPG/PPG to HbA1c
Optimal glycemic control is paramount in diabetes management. Measurement of glycated hemoglobin (HbA1c), fasting plasma glucose (FPG), and postprandial plasma glucose (PPG) assesses glucose control. Although HbA1c is the gold standard, its cost limits accessibility in resource-constrained settings. In such cases, estimating postprandial and fasting glucose gains importance, especially in developing countries, to evaluate glycemic control19.
In diabetes screening, dipstick kits are initially used for qualitative assessment of glucosuria, but their lack of quantitative precision necessitates confirmation through laboratory testing of urine glucose and HbA1c levels. These quantitative measures provide more accurate insights into glycemic control and guide health care providers in diagnosis, treatment, and monitoring strategies for diabetes management. Therefore, while dipstick kits serve as valuable initial indicators, reliance on laboratory reports ensures thorough evaluation and appropriate intervention in diabetes care20.
Lifestyle Factors
Nepal’s predominant agrarian nature entails a significant portion of its population engaging in labor-intensive occupations. The national living standard survey highlights this by revealing that approximately 80% of Nepal’s populace resides in rural regions, where limited access to motor vehicle transportation infrastructure makes walking a prevalent mode of daily commuting21.
Considering the diverse challenges posed by diabetes management and the extensive applications of insulin therapies, the following patient-centered recommendations have been formulated to guide health care professionals in navigating the complexities of treatment.
Expert Recommendations
Management of Diabetes has Moved from Glucocentrica to Patient Centric Approach
Panel Discussion: Insulin therapy plays a crucial role in maintaining glycemic control. According to experts, premixed insulins, favored for their balance of effectiveness and patient convenience, are particularly suitable for Asian patients with high-carbohydrate diets. Analog premixed variants offer faster onset and extended duration compared to conventional versions. Human insulin 70/30 and Lispro mix 25 provide flexibility and prolonged action. Insulin glargine stands out for its consistent 24-hour activity with a single injection, making it the most prescribed long-acting insulin analogue, offering patients safe, effective, and potentially cost-efficient treatment options.
Consensus 1: A person-centered approach should be used to optimize management of diabetes.
Evidence: Choosing pharmacologic agents for T2DM management should prioritize a person-centered approach, considering individual factors. For those with fasting hyperglycemia, bedtime basal insulin or premixed injections before dinner are options, while postprandial hyperglycemia may benefit from a breakfast dose. Consistent high glycemic levels may require a two- or three-dose premixed regimen influenced by dietary habits. High-carb meal consumers may respond better to high-mix formulations, while those prone to hypoglycemia may prefer low-mix formulations, especially analogues. Counseling individuals on premixed insulin should include advice to avoid vigorous physical activity within 2-3 hours of injection22,23.
A systemic review indicated similar clinical efficacy and safety of Glargine compared to its reference products, making biosimilars viable alternatives for insulin therapy in both type 1 and type 2 diabetes patients24,25. Evidence suggests that increased use of biosimilar insulin may reduce costs for consumers, with a Canadian study projecting substantial potential savings compared to the originator. Furthermore, the introduction of biosimilar insulin was linked to reduced reimbursement, indicating increased market competition and lower insulin costs26.
Choice of Insulin Based on Glycemic Parameters
Panel discussion: Basal insulin effectively controls FBG levels, while rapid-acting insulin is necessary for elevated PPG levels. As basal insulin aligns with FBG control, FBG levels can be an objective marker for insulin therapy determination27. A high FBG level suggests a basal insulin regimen, whereas relatively normal FBG levels, despite uncontrolled HbA1c, may prompt consideration of alternative treatments. However, relying solely on FBG has limitations as it doesn't assess postprandial hyperglycemia. When choosing between basal and basal-bolus insulin regimens, FBG, PPG, and HbA1c should be considered28. With its safer peak-less glycemic profile, insulin glargine is suitable for aggressive treatment regimens, potentially helping more patients achieve tight glycemic control as current guidelines recommend. (Table 3)
Consensus 2: Insulin therapy to be considered at any stage under specific circumstances. Periodic evaluation of clinical factors is crucial before initiating and titrating insulin to mitigate the risk of hypoglycemia.
Evidence: Basal insulin is more effective in managing fasting glycemia than prandial insulin, aligning with its fundamental pharmacology. Elevated FPG levels are recommended for prescribing basal insulin, whereas relatively normal FPG levels amidst uncontrolled HbA1c might suggest alternative approaches. High PPG levels indicate rapid-acting insulin requirement integrated into premixed, prandial, or basal-bolus regimens. Timing injections based on PPG peaks after meals can guide treatment. For instance, once-daily premixed insulin aligns with the meal, causing the highest PPG surge. Similar considerations apply to the bolus component of basal plus regimes. Basal-bolus protocols suit 'very high' HbA1c levels, while lower-dose regimens suffice for 'less high' HbA1c cases29.
In a randomized controlled trial on T2DM patients, researchers compared once-daily and twice-daily intermediate-acting insulin (Neutral Protamine Hagedorn
|
Table 3. Choice of Insulin Based on Clinical Factors
|
|
Clinical factor
|
Basal
|
Premix
|
Intensive
|
|
Fasting hyperglycemia
|
++
|
+
|
++
|
|
Postprandial hyperglycemia
|
-
|
+
|
++
|
|
Both fasting and postprandial hyperglycemia
|
-
|
++
|
++
|
|
High HbA1c (>8.5%)
|
-
|
++
|
++
|
|
Low HbA1c (<8.5%)
|
+
|
++
|
-
|
HbA1c: Hemoglobin A1c
insulin) with or without oral drugs. Subjects with a ratio of ≥1.3 (observed in 60% of participants), indicative of fasting hyperglycemia dominance, exhibited similar responses across all four studied regimes. Those with a lower ratio (<1.3), suggestive of overall hyperglycemia, responded better to twice-daily insulin30.
Strategies for Selection of Insulin in Nepalese Patients
SECURE Model
Panel discussion: The SECURE model highlights six critical factors for personalized glucose-lowering treatment and glycemic targets. Insulin analogs, like Lispro and glulisine, improve postprandial glucose control and reduce hypoglycemia risk in CKD patients. Dosing varies for different insulins based on estimated glomerular filtration rate (eGFR) stages, with glargine showing a lower hypoglycemia risk than NPH insulin. Detemir offers continuous coverage with twice-daily dosing, while newer glargine formulation minimizes hypoglycemia. Degludec insulin presents potential cognitive benefits with fewer hypoglycemic incidents. Overall, long-acting analogs detemir and glargine are preferred for better control and lower severe hypoglycemia, while human insulins suit those with cost constraints. Over the last decade, insulin glargine has become a standard of care in diabetes treatment in Nepal due to its well-established safety and efficacy profiles. (Table 4)
Consensus 3: SECURE model proposes a holistic and comprehensive approach to hyperglycemia management particularly in ill patients.
Evidence: Insulin analogs, such as lispro and glulisine, demonstrate reduced hypoglycemia risk and improved postprandial glucose control, which is particularly beneficial for chronic kidney disease (CKD) patients due to faster absorption31. Comparing prandial insulins, both lispro, and glulisine effectively suppressed postprandial hyperglycemia in comparison to regular insulin.
The dosing of aspart did not exhibit significant differences across various stages of eGFR (<60 mL/min, 60-80 mL/min, >90 mL/min). However, there was a notable decrease in lispro and human insulin doses among patients with eGFR <60 mL/min32. Insulin glargine demonstrates a reduced risk of nocturnal and overall hypoglycemia compared to NPH insulin, primarily attributed to the peak in action occurring 4-10 hours after NPH insulin administration. Nonetheless, NPH insulin remains a cost-effective choice for basal insulin33.
Insulin detemir, at lower doses, requires twice-daily dosing for continuous coverage with no significant peak in action. The newer formulation of insulin glargine (300 units/mL) shows a lower hypoglycemia risk compared to the U-100 glargine (100 units/mL) formulation34,35. Human insulins offer advantages for individuals with cognitive impairment and are suitable for those pursuing less intensive A1c goals or facing insulin resistance and cost concerns. This is particularly relevant for type 1 diabetes patients who may not afford insulin analogs, including biosimilars36.
Biopsychosocial Model
Panel discussion: Flexibility in insulin management, in line with the biopsychosocial model of health care, considers the individual's physical, psychological, and social aspects. Basal insulins, like glargine and degludec, offer simplicity and flexibility with one injection per day, allowing lifestyle freedom. Rapid-acting analogs provide greater flexibility with a shorter injection-meal gap.
Dual-action insulins, such as premixed insulins, offer some flexibility in injection timing but may have limitations in dose titration. Mastering basic skills enables the flexible use of premixed insulins despite their complexity. A number-based taxonomy approach encompasses traditional and newly developed insulin regimes, providing a comprehensive framework for classification. (Table 5)
Consensus 4: Tailoring insulin regimens to individual lifestyles optimizes diabetes management and enhances treatment adherence.
Evidence: Studies have shown that people with diabetes face various challenges in managing their condition and desire greater involvement in their care. Flexibility in insulin management responds to patient needs and convenience, consistent with the principles of person-centered care, which aims to tailor treatment plans to the individual's unique circumstances and preferences37. Basal insulin regimens are considered
|
Table 4. Choice of Insulin Based on the SECURE Model
|
|
SECURE Model
|
Choice of insulin
|
Benefits
|
|
Severity
|
Analogue insulin (Degludec + Aspart, Aspart)
|
Low-risk of hypoglycemia in CKD patients
Better PPG control with faster absorption
|
|
Rapid-acting
(Lispro, Glulisine)
|
No change in PK parameters in CKD patients
|
|
Basal insulin
(Glargine)
|
Rapid HbA1c reduction, stable half-life, and longer duration of action in patients with renal failure
|
|
Expected prognosis
|
Basal insulin (Glargine, Detemir, Degludec)
|
Choice for obese patients with T2DM
|
|
Concomitant medication
|
Short-acting insulin (Regular human)
|
In patients with significant hyperglycemia or impaired health status after GC administration
|
|
Rapid-acting insulin
(Lispro, Glulisine, Aspart)
|
Hydrocortisone is usually administered twice or thrice daily, (multiple doses might be suitable to improve glycemic control)
|
|
Urgency of control
|
Basal bolus (Glargine, Detemir, Degludec)
|
It can be given to patients with type 1 diabetes or life-, organ-, or limb-threatening complications
|
|
Risk of hypoglycemia
|
Long-acting insulin
(Detemir, Glargine)
|
In patients with more advanced cognitive dysfunction or dementia, it may be best to implement a regimen using a dose that will not cause hypoglycemia combined with conservative fixed mealtime doses that are given immediately after a patient has eaten an adequate meal.
|
|
Environmental factors
|
Long-acting insulin (Detemir, Glargine)
|
Both insulin detemir and glargine are cost-effective compared to NPH insulin for T2DM patients, especially when the benefit of reducing the hypoglycemia event rate is considered.
|
CKD: Chronic kidney disease; GC: Glucocorticoids; NPH: Neutral protamine hagedorn; PPG: Postprandial glucose; T2DM: Type 2 diabetes mellitus.
|
Table 5. Choice of Insulin Based on Psychosocial Factors
|
|
Psychosocial factor
|
Basal
|
Premix
|
Intensive
|
|
Inability to have regular meal
|
+
|
+
|
-
|
|
Inability to self-monitor/self-administration
|
+
|
+
|
-
|
|
Inability to remain in regular touch with the diabetes care team
|
+
|
+
|
-
|
|
Psychosocial factors
|
+
|
+
|
-
|
|
Poor family support and acceptance
|
+
|
+
|
-
|
|
Low personal acceptance of insulin
|
+
|
+
|
-
|
for their simplicity, minimal intrusion, and adaptability. They involve just one daily injection, don't necessitate strict meal adherence, and offer lifestyle flexibility38. While Neutral Protamine Hagedorn insulin is best taken with a snack, basal analogs can be administered without considering mealtimes. Human regular insulin should be given 30 minutes before a meal, whereas rapid-acting analogs like aspart, glulisine, and lispro offer a shorter injection-meal gap and can be injected
5 minutes before or after a meal without compromising effectiveness. Human premixed insulins require a 30-minute premeal injection, while premixed insulin analogs can be taken 5 minutes before or immediately after a meal39. Insulin coformulations, such as human insulin 30/70 and lispro mix 25, offer flexibility in adjusting administration timing37,40.
Choice of Insulin in Special Condition
Panel discussion: In critically ill patients, continuous intravenous insulin is preferred, while noncritically ill patients with regular meals may receive basal and correction insulin doses. A basal plus bolus correction insulin regimen is recommended for noncritically ill patients with good nutritional intake. Sliding scale insulin is advised against in the inpatient hospital setting. Basal insulin is widely recommended for transitioning from intravenous to subcutaneous insulin therapy and maintaining glucose control. Once-daily glargine insulin offers practicality and simplicity, making it a convenient initiation strategy. Glargine insulin suits various treatment intensities, providing
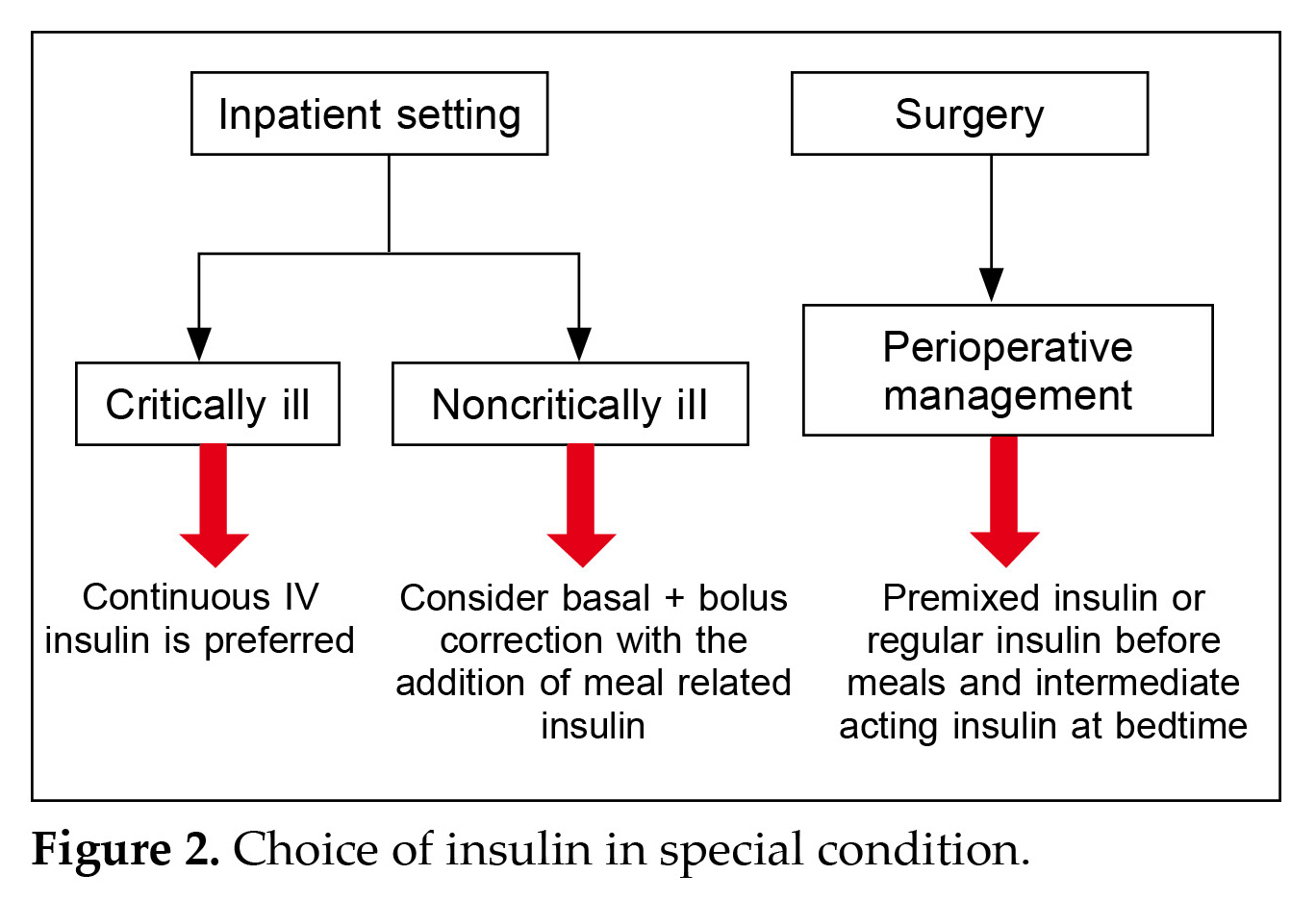
regimen flexibility for patient convenience. Early insulin introduction in type 2 diabetes patients is encouraged. (Fig 2)
Consensus 5: In both critically ill and noncritically ill patients, when blood glucose levels reach 80 mg/dL, initiating insulin therapy is essential for addressing persistent hyperglycemia.
Evidence: For noncritically ill patients with good nutritional intake, a combination of basal, meal-related, and correction insulin doses is preferred. Subcutaneous rapid- or short-acting insulin can be administered before meals or every 4-6 hours to correct hyperglycemia in patients not on regular meals or receiving continuous enteral/parenteral nutrition, for noncritically ill patients with poor oral intake or those not taking anything by mouth, basal insulin or basal plus bolus correction insulin regimen is preferred over sliding scale insulin due to better glycemic control and reduced hospital complications. In the outpatient setting, premixed insulin is preferred, while in the inpatient setting, basal-bolus therapy is recommended. When transitioning patients with T1DM or T2DM to the outpatient setting, subcutaneous insulin should be started, with basal insulin initiated 2-4 hours before discontinuing IV insulin to facilitate a smooth transition29. For patients with T1DM and T2DM undergoing surgery, long-acting insulin (glargine) should be discontinued 2-3 days before the procedure. For glycemic control during the perioperative period, a combination of intermediate-acting insulin (NPH) with short—or rapid-acting insulin administered twice daily or regular insulin before meals, along with intermediate-acting insulin at bedtime41,42.
Storage of Insulin Based on Geographical Conditions in Nepal
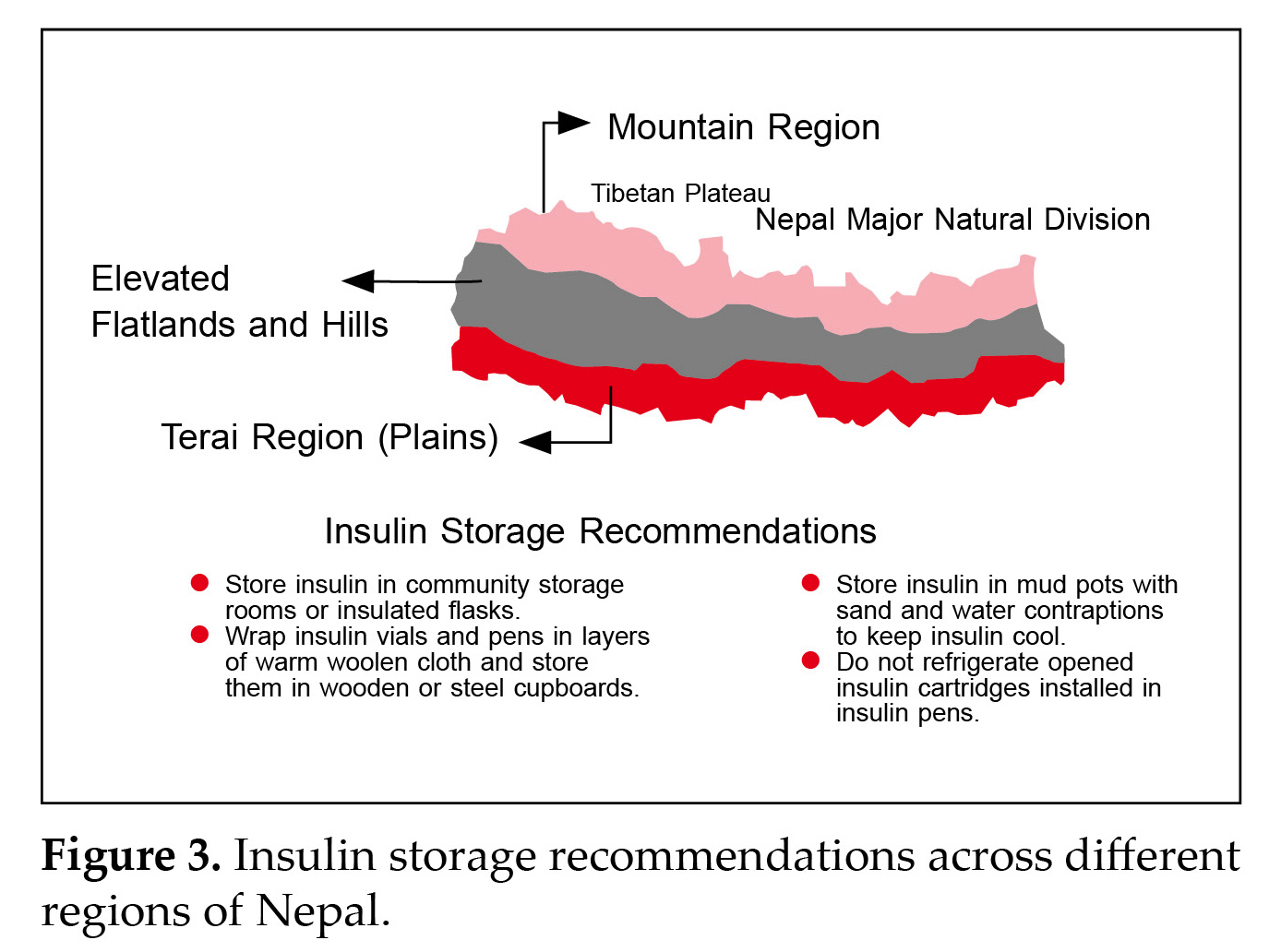
Panel discussion: Addressing the unique challenges of insulin storage in Nepal necessitates locally relevant and improvised solutions. Ideally, insulin should be stored in a refrigerator between 2 and 8°C and protected from light when unopened. In hot regions like Terai, mud pots with sand and water contraptions can keep insulin cool, while in cold areas, community storage rooms or insulated flasks are suitable. Patients are advised to wrap insulin in warm woolen cloth and store it in wooden or steel cupboards, considering frequent power cuts. Opened vials can be stored at room temperature (15-30°C) for 4 to 6 weeks or in a refrigerator (2-8°C) until expiry, but insulin should never be frozen. (Fig 3)
Limited access to medical services and dietary patterns poses challenges at higher altitudes. Extreme temperatures and difficulty in accessing and storing insulin are concerns. Various items like abdominal binders made from wool and yak, repurposed transceiver bags, homemade fleece bags, and foam pouches are used for storage and transport. Altitude may affect glycemic control, causing delayed carbohydrate absorption and potential postprandial hypoglycemia above 5000 meters. Adjustments to insulin dosages may be needed due to carbohydrate-rich diets. Glucometer readings may be slightly inaccurate at high altitudes, but their clinical significance is minimal.
Consensus 6: Proper storage of insulin is essential for Nepalese patients to ensure effective management of diabetes.
Evidence: In Nepal, where outdoor temperatures drop as low as -30°C and indoor temperatures vary between 4 to 20°C due to heating methods like burning iron stoves, maintaining appropriate storage conditions for insulin becomes a significant concern since access to health care facilities and supplies is limited. Extreme heat in living rooms and freezing temperatures in adjacent rooms make it challenging to find suitable storage locations. In warmer regions of Nepal like the Terai, where room temperatures often go above 25°C, insulin storage is a concern. Studies indicate that storing insulin (regular and biphasic) at 32°C and 37°C for 28 days leads to a 14-18% potency decrease. Moreover, it's advised not to refrigerate opened insulin cartridges installed in insulin pens43,44.
Disposal Strategies for Insulin
Panel discussion: Proper disposal of needles and cartridges aligns with the National biomedical waste guidelines for responsible medical waste management, ensuring safe practices. Glucose monitoring sticks and similar solid waste items are disposed of in yellow non-chlorinated bags, offering flexibility while maintaining safety. Disposable insulin pens, especially after removing pen needles, should be appropriately disposed of to ensure individual safety and proper medical waste handling. Transportation of biomedical waste strictly follows guidelines, using
authorized vehicles and safety protocols to minimize risks. To ensure timely and secure handling, storage of biomedical waste items, including those contaminated with blood and body fluids, should not exceed 48 hours without treatment.
Implementation of these measures ensures safe and effective insulin management, considering Nepal's environmental conditions and resources. Needles can be collected in narrow-mouth containers to prevent children from accessing them and discarded in biomedical waste centers. Patients should receive awareness regarding the disposal of single-use pens, cartridges, and needles from health care providers. Adopting vaccine vial monitor (VVM) technology is crucial for safeguarding insulin quality, as it changes color if a specific threshold is reached.
Consensus 7: Proper insulin disposal strategies are essential for Nepalese patients to safeguard public health and the environment.
Evidence: A survey in Nepal highlights concerning trends among diabetes patients, with many failing to adhere to proper insulin-related waste disposal practices. Approximately half of the surveyed participants reported disposing of used needles in bins, while others resorted to discarding them in isolated areas or burning them.
One patient even stored used needles in a plastic container inside their refrigerator for 3 months. These findings underscore the urgent need for enhanced awareness and education on safe insulin disposal methods in the Nepalese diabetes patient community45.
Additionally, Nepalese patients often perceive insulin treatment as a last resort. Improper disposal practices for used needles were widespread, including handing them over to municipal waste disposal services or discarding them in isolated locations. Lack of awareness and regulatory requirements contribute to these practices46.
Exposure to high temperatures can reduce insulin potency, with storage at 32-37°C causing a 14-18% potency loss. It's suggested that insulin manufacturers adopt cost-effective technology using VVM. It is a thermochromic label on vaccine vials that changes color to indicate temperature exposure beyond recommended levels, jeopardizing vaccine potency.
This technology, effective in the polio eradication program in India, can be scaled up, benefiting insulin storage and preserving its effectiveness, as recognized by the World Health Organization47.
Diabetes Awareness and Prevention Strategies
Strategies beyond diabetes education are recommended to address insulin distress and improve self-management practices. Strengthening communication between physicians and patients, enhancing coping skills, and implementing motivational measures are suggested. These recommendations aim to enhance diabetes self-management, provide education and counseling, and offer emotional support to individuals with diabetes48,49. (Table 6 and 7)
Final Consensus Statements
- A person-centered approach should be used to optimize management of diabetes.
- Blood glucose monitoring is an integral part of insulin therapy, providing essential guidance for tailoring regimens.
- SECURE model proposes a holistic and comprehensive approach to hyperglycemia management particularly in ill patients.
- Tailoring insulin regimens to individual lifestyles optimizes diabetes management and enhances treatment adherence.
- In both critically ill and non-critically ill patients, when blood glucose levels reach 180 mg/dL, initiating insulin therapy is essential for addressing persistent hyperglycemia.
- Proper storage of insulin is essential for Nepalese patients to ensure effective management of diabetes.
- Proper insulin disposal strategies are essential for Nepalese patients to safeguard public health and the environment.
|
Table 6. Patient and Physician-oriented Strategies for Awareness and Prevention of Diabetes
|
|
Parameter
|
Physician-oriented
|
Patient-oriented
|
|
Awareness
|
Organize medical conferences or workshops to update physicians about the latest evidence-based guidelines and advancements in insulin therapy.
|
Conduct educational campaigns to increase awareness among patients about the importance of insulin therapy for diabetes management.
|
|
Adoption
|
Encourage physicians to proactively discuss and recommend insulin therapy to eligible patients, highlighting its advantages and addressing concerns.
|
Provide easily understandable information to patients about the benefits and necessity of insulin therapy, addressing misconceptions and fears.
|
|
Accessibility
|
Work with health care systems and policymakers to improve the availability of insulin in health care facilities and enhance distribution networks.
|
Advocate for improved accessibility of insulin, including availability in pharmacies, hospitals, and remote areas, to ensure patients can access it conveniently.
|
|
Affordability
|
Advocate physicians in identifying alternative insulin options or assistance programs for patients who face financial challenges.
|
Collaborate with insurance companies or government agencies to make insulin more affordable through subsidies, insurance coverage, or price reduction programs.
|
|
Acceptability
|
Offer continuing medical education programs to enhance insulin therapy and pen use understanding.
|
Demonstrate proper injection techniques and provide tools like pen injectors to make administration easier.
Collect patient feedback to identify barriers and opportunities for improvement in insulin acceptability and adherence.
|
|
Table 7. Practical Guidance for Choice of Insulin
|
|
Parameter
|
Patient profile
|
Choice of insulin
|
| |
|
Basal (Glargine, detemir, degludec, neutral protamine hagedorn (NPH) insulin)
|
Prandial (Aspart, glulisine, lispro, regular insulin)
|
|
Duration of diabetes/ uncontrolled hyperglycemia
|
Longer duration of diabetes and uncontrolled hyperglycemia
|
a ± a
|
|
Symptoms of hyperglycemia
|
Persons with symptomatic diabetes (polyuria, polydipsia, polyphagia, weight loss, frequent infection)
|
a ± a
|
|
Lifestyle and meal pattern*
|
High postprandial glucose levels due to the intake of high-carbohydrate meals in large quantity
|
|
a
|
|
Obese Patient with T2DM
|
a
|
|
|
Risk of hypoglycemia
|
CKD patient with T2DM
|
a ± a
|
| |
Advanced cognitive dysfunction or dementia
|
a
|
|
|
Urgency of control
|
Diabetic patient with life-, organ-, or limb-threatening complication
|
a
|
|
|
Combination therapy
|
Inadequacy of multiple drugs that target postprandial glycemia, e.g., sulfonylureas and alpha-glucosidase inhibitors
|
|
a
|
| |
Inadequacy of drugs that target both fasting and postprandial glycemia, e.g., DPP4i, GLP1RA and SGLT2i
|
a ± a
|
| |
Inadequacy of basal insulin
|
a
|
| |
|
|
Add prandial insulin separately or as part of a dual-action insulin
|
| |
Inadequacy of once-daily premixed insulin
|
Twice daily or more frequent insulin administration
|
|
In patient setting
|
Critically ill
|
Continuous IV insulin is preferred
|
| |
Noncritically ill
|
Basal + Bolus correction with addition of meal related insulin
|
|
Surgery
|
Perioperative management
|
Premixed insulin before meals, intermediate acting insulin at bedtime
|
*Meal pattern (number of meals or snacks per day), relative quantity of meals, their composition (proportion of carbohydrates, glycemic index), and regularity
CKD: Chronic kidney disease; DPP4i: Dipeptidyl peptidase-4 inhibitor; GLP1RA: Glucagon-like peptide-1 receptor agonist; IV: intravenous;
SGLT2i: Sodium-glucose cotransporter-2 inhibitor; T2DM: Type 2 diabetes mellitus.
Conclusion
Addressing the complex landscape of diabetes management in Nepal requires a multifaceted approach that acknowledges the country's diverse topography, cultural intricacies, and health care resource limitations. The consensus reached through collaborative efforts among health care professionals, guided by patient-centered principles, is crucial for optimizing insulin therapy and improving diabetes outcomes.
By recognizing the importance of effective communication, tailored treatment regimens, and proper utilization of insulin, health care providers can navigate the challenges posed by diabetes in Nepal more effectively. Moreover, the inclusion of diverse stakeholders, including patients, caregivers, policymakers, and civil society, is essential for implementing comprehensive strategies that address the unique needs of Nepalese communities.
Continued efforts to enhance awareness, promote evidence-based practices, and strengthen health care infrastructure will be pivotal in overcoming barriers to diabetes management in Nepal. Through collective agreement and concerted action, we can strive towards better patient outcomes, improved public health, and a more sustainable approach to diabetes care in Nepal and beyond.
References
- The IDF Diabetes Atlas 10th Edition. Available from: https://diabetesatlas.org/
- Diabetes in SEA. Available from: https://www.idf.org/our-network/regions-members/south-east-asia/diabetes-in-sea.html
- Bhandari GP, Angdembe MR, Dhimal M, Neupane S, Bhusal C. State of non-communicable diseases in Nepal. BMC Public Health. 2014;14:23.
- Geography of Nepal. Embassy of Nepal - Tokyo, Japan. Available from: https://jp.nepalembassy.gov.np/geography-of-nepal/
- Shrestha N, Karki K, Poudyal A, Aryal KK, Mahato NK, Gautam N, et al. Prevalence of diabetes mellitus and associated risk factors in Nepal: findings from a nationwide population-based survey. BMJ Open. 2022;12(2):e060750.
- Pandey AR, Aryal KK, Shrestha N, Sharma D, Maskey J, Dhimal M. Burden of diabetes mellitus in Nepal: an analysis of global burden of disease study 2019. J Diabetes Res. 2022:2022:4701796.
- Kalra S, Megallaa MH, Jawad F. Perspectives on patient-centered care in diabetology. J Midlife Health. 2012;3(2):
93-6.
- Kalra S, Baruah MP, Kalra B. Diabetes care: evolution of philosophy. Indian J Endocrinol Metab. 2017;21(4):495-7.
- Kalra S, Verma M, Kapoor N. The quintessential quincunx: a model for health care delivery. J Pak Med Assoc. 2022;72(3):572-3.
- Sapkota S, Brien JAE, Aslani P. Nepalese patients’ perceptions of treatment modalities for type 2 diabetes. Patient Prefer Adherence. 2016;10:1777-86.
- Sapkota S, Brien JAE, Aslani P. Nepalese patients’ anti-diabetic medication taking behaviour: an exploratory study. Ethn Health. 2018;23(7):718-36.
- Blonde L. Current challenges in diabetes management. Clin Cornerstone. 2005;7 Suppl 3:S6-17.
- Janež A, Guja C, Mitrakou A, Lalic N, Tankova T, Czupryniak L, et al. Insulin therapy in adults with type 1 diabetes mellitus: a narrative review. Diabetes Ther. 2020;11(2):387-409.
- Practice Guidelines Resources | American Diabetes Association. Available from: https://professional.diabetes.org/content-page/practice-guidelines-resources
- Samson SL, Vellanki P, Blonde L, Christofides EA, Galindo RJ, Hirsch IB, et al. American Association of Clinical Endocrinology Consensus Statement: Comprehensive Type 2 Diabetes Management Algorithm – 2023 Update. Endocr Pract. 2023;29(5):305-40.
- Joshi A, Adhikari B, Tamrakar B, Puppalwar G, Shah A, Jain R. Assessment of safety and effectiveness of glaritus® (Wockhardt’s insulin glargine) in a prospective, multi-centric post marketing observational study in Nepalese having type 2 diabetes mellitus. Open J Endocrine Meta Dis. 2019;9(2):21-33.
- Swinnen SG, Hoekstra JB, DeVries JH. Insulin therapy for type 2 diabetes. Diabetes Care. 2009;32(Suppl 2):S253-9.
- Shrestha A. Diet, obesity, and diabetes in suburban Nepal - a community-based study. 2015. Available from: https://digital.lib.washington.edu:443/researchworks/handle/1773/33885
- Ketema EB, Kibret KT. Correlation of fasting and postprandial plasma glucose with HbA1c in assessing glycemic control; systematic review and meta-analysis. Arch Public Health. 2015;73:43.
- Agbozo F, Abubakari A, Narh C, Jahn A. Accuracy of glycosuria, random blood glucose and risk factors as selective screening tools for gestational diabetes mellitus in comparison with universal diagnosing. BMJ Open Diabetes Res Care. 2018;6(1):e000493.
- Central Bureau of Statistics – Central Bureau of Statistics. Available from: https://nsonepal.gov.np/
- Kalra S, Czupryniak L, Kilov G, Lamptey R, Kumar A, Unnikrishnan AG, et al. Expert opinion: patient selection for premixed insulin formulations in diabetes care. Diabetes Ther. 2018;9(6):2185-99.
- Kalra S, Plata-Que T, Kumar D, Mumtaz M, Søndergaard F, Kozlovski P, et al. Initiation with once-daily BIAsp 30 results in superior outcome compared to insulin glargine in asians with type 2 diabetes inadequately controlled by oral anti-diabetic drugs. Diabetes Res and Clin Pract. 2010;88(3):282-8.
- Tieu C, Lucas EJ, DePaola M, Rosman L, Alexander GC. Efficacy and safety of biosimilar insulins compared to their reference products: a systematic review. PLoS ONE. 2018;13(4):e0195012.
- Saunders H, Pham B, Loong D, Mishra S, Ashoor HM, Antony J, et al. The cost-effectiveness of intermediate-acting, long-acting, ultralong-acting, and biosimilar insulins for type 1 diabetes mellitus: a systematic review. Value Health. 2022;25(7):1235-52.
- Developing more biosimilar insulins could reduce costs for people with diabetes. Available from: https://www.healio.com/news/endocrinology/20220607/developing-more-biosimilar-insulins-could-reduce-costs-for-people-with-diabetes
- Lasserson DS, Glasziou P, Perera R, Holman RR, Farmer AJ. Optimal insulin regimens in type 2 diabetes mellitus: systematic review and meta-analyses. Diabetologia. 2009;52(10):1990-2000.
- Kalra S, Gupta Y. Insulin initiation: bringing objectivity to choice. J Diabetes Metab Disord. 2015;14:17.
- Silver B, Ramaiya K, Andrew SB, Fredrick O, Bajaj S, Kalra S, et al. EADSG Guidelines: Insulin Therapy in Diabetes. Diabetes Ther. 2018;9(2):449-92.
- Vähätalo M, Rönnemaa T, Viikari J. Recognition of fasting or overall hyperglycaemia when starting insulin treatment in patients with type 2 diabetes in general practice. Scand J Prim Health Care. 2007;25(3):147-53.
- Iglesias P, Díez JJ. Insulin therapy in renal disease. Diabetes Obes Metab. 2008;10(10):811-23.
- Ruggenenti P, Flores C, Aros C, Ene-Iordache B, Trevisan R, Ottomano C, et al. Renal and metabolic effects of insulin lispro in type 2 diabetic subjects with overt nephropathy. Diabetes Care. 2003;26(2):502-9.
- Abbatecola AM, Bo M, Barbagallo M, Incalzi RA, Pilotto A, Bellelli G, et al. Severe hypoglycemia is associated with antidiabetic oral treatment compared with insulin analogs in nursing home patients with type 2 diabetes and dementia: results from the DIMORA study. J Am Med Dir Assoc. 2015;16(4):349.e7-12.
- Riddle MC, Bolli GB, Ziemen M, Muehlen-Bartmer I, Bizet F, Home PD, et al. New insulin glargine 300 units/mL
versus glargine 100 units/mL in people with type 2 diabetes using basal and mealtime insulin: glucose control and hypoglycemia in a 6-month randomized controlled trial (EDITION 1). Diabetes Care. 2014;37(10):2755-62.
- Dzygalo K, Golicki D, Kowalska A, Szypowska A. The beneficial effect of insulin degludec on nocturnal hypoglycaemia and insulin dose in type 1 diabetic patients: a systematic review and meta-analysis of randomised trials. Acta Diabetol. 2015;52(2):231-8.
- Herman WH, Kuo S. 100 years of insulin: Why is insulin so expensive and what can be done to control its cost? Endocrinol Metab Clin North Am. 2021;50(3 Suppl):e21-34.
- Kalra S, Gupta Y, Unnikrishnan AG. Flexibility in insulin prescription. Indian J Endocrinol Metab. 2016;20(3):408-11.
- Kalra S. Basal insulin analogues in the treatment of diabetes mellitus: what progress have we made? Indian J Endocrinol Metab. 2015;19(Suppl 1):S71-3.
- Bilotta F, Guerra C, Badenes R, Lolli S, Rosa G. Short acting insulin analogues in intensive care unit patients. World J Diabetes. 2014;5(3):230-4.
- Kalra S, Gupta Y. Number-based approach to insulin taxonomy. Diabetes Ther. 2015;6(4):469-79.
- Meneghini LF. Perioperative management of diabetes: translating evidence into practice. Cleve Clin J Med. 2009;76 Suppl 4:S53-9.
- Moghissi ES, Korytkowski MT, DiNardo M, Einhorn D, Hellman R, Hirsch IB, et al. American Association of Clinical Endocrinologists and American Diabetes Association consensus statement on inpatient glycemic control. Diabetes Care. 2009;32(6):1119-31.
- Khurana G, Gupta V. Effect on insulin upon storage in extreme climatic conditions (Temperature and pressure) and their preventive measures. J Soc Health Diab. 2019;7:6-10.
- Mokta JK, Kalra S. Insulin storage in the Upper Himalayas. Rural Remote Health. 2014;14(3):2983.
- Poudel RS, Shrestha S, Piryani RM, Basyal B, Kaucha K, Adhikari S. Assessment of insulin injection practice among diabetes patients in a tertiary healthcare centre in Nepal: a preliminary study. J Diabetes Res. 2017;2017:8648316.
- Patil M, Sahoo J, Kamalanathan S, Selviambigapathy J, Balachandran K, Kumar R, et al. Assessment of insulin injection techniques among diabetes patients in a tertiary care centre. Diabetes Metab Syndr. 2017;11 Suppl 1:S53-56.
- Balachandran K. Cold chain and insulin: should we follow the polio vaccine example? Indian J Endocrinol Metab. 2019;23(3):379.
- Adhikari M, Devkota HR, Cesuroglu T. Barriers to and facilitators of diabetes self-management practices in Rupandehi, Nepal-multiple stakeholders’ perspective. BMC Public Health. 2021;21(1):1269.
- American Diabetes Association. Standards of Medical Care in Diabetes—2010. Diabetes Care. 2010;33(Suppl 1):S11-61.