Abstract
Background: Diabetes is a progressive disease managed by different oral antidiabetic drugs (OADs) with or without glimepiride/metformin. As diabetes continues to be a significant health concern in India, novel therapeutic strategies are essential to effectively control the disease and improve patient outcomes. New drugs like sodium-glucose cotransporter-2 inhibitors (SGLT2i) and dipeptidyl peptidase-4 inhibitors (DPP-4i) have intermediate efficacy. Understanding clinicians’ prescription patterns is crucial for optimizing treatment strategies for better long-term type 2 diabetes mellitus (T2DM) control. Methods: This was a retrospective, multicenter, observational case-based questionnaire study on T2DM patients undergoing pharmacotherapy. It aimed to collect data on clinical utilization patterns of glimepiride and metformin FDC (fixed-dose combination) with other OADs and comorbidities. The study included responses from 500 health care professionals (HCPs) across India. Statistical analysis was performed using SPSS® Version 23.0 software. Independent t-test was used to compare the change in fasting plasma glucose (FPG), postprandial plasma glucose (PPG), and glycated hemoglobin (HbA1c) between two groups and Fisher’s exact and Chi-square tests were used to compare categorical variables. P-value <0.05 was considered statistically significant. Results: The study analysis included responses from 500 HCPs. It showed that 6,250 patients received glimepiride/metformin FDC. The HbA1c was found to be 8.81% before treatment, which decreased to 7.75% after treatment. Among the 6,250 patients, 1,704 patients also recieved other OADs, where some patients recieved more thn one OADs. DPP4i was prescribed the most (1,064 patients followed by sodium-glucose cotransporter 2 inhibitors (SGLT2i) (573 patients), pioglitazone (229 patients), alpha-glucosidase inhibitor (AGI) (207 patients), insulin (178 patients), and lastly glucagon-like peptide 1 receptor agonist (GLP1RA) being prescribed in 35 patients along with the combination. Hypoglycemia was observed in very few patients (4.49%). Hypertension was the most prevalent (60.5%) comorbidity in the studied patient population. Conclusion: Use of glimepiride and metformin FDC along with other OADs offer optimized glycemic control, promote weight loss, and help to reduce complications in patients with T2DM.
Keywords: Type 2 diabetes mellitus, OADs, glimepiride, metformin, glycemic control, HbA1c
Introduction
Diabetes has emerged as a global epidemic, affecting millions of people worldwide1. The incidence of diabetes has been increasing in South-East Asian countries for at least 20 years, according to the International Diabetes Federation (IDF) 10th edition, and current figures have surpassed all prior projections.
According to a recent study by Kumar et al the prevalence of diabetes was 10.5%, 8.8%, and 9.6%, respectively, in the globe, Southeast Asia, and India in 2021, and it will increase to 12.5%, 11.5%, and 10.9%, respectively, by 20452.
India, in particular, has witnessed a significant rise in the prevalence of diabetes over the past few decades, making it a major public health concern. Over 77 million people in India are dealing with diabetes. By 2045, researchers project that number will rise to 134 million3.
Managing diabetes effectively requires a comprehensive approach, as shown in Figure 1.
Among the various treatment options available, oral antidiabetic drugs (OADs) play a crucial role in controlling blood glucose levels and preventing complications. Accordingly, metformin is the recommended first-line diabetes treatment option4.
The use of modern sulfonylureas (SUs) like glimepiride in type 2 diabetes mellitus (T2DM) management has been advocated by a number of other international organizations, including the World Health Organization (WHO), South Asian Federation of Endocrine Societies (SAFES), IDF, and American Diabetes Association/European Association5-8. The WHO advises using SUs in combination with the first-line therapy among patients who are unable to achieve treatment objectives with first-line oral hypoglycemic medications9.
Additionally, the modern SUs have significant safety and efficacy profile. A study by Basit et al (2012) have shown that glimepiride is a safer and more affordable option for treating T2DM, as it lowers fasting blood sugar, post-meal glucose, and glycated hemoglobin (HbA1c) levels, without adversely affecting ischemic preconditioning10. The evidence of glimepiride’s cardiovascular safety from the CAROLINA trial, compared to dipeptidyl peptidase-4 inhibitors (DPP4i), will provide cardiologists with greater confidence to use it in various conditions, including stable coronary artery disease, cerebrovascular disease, and peripheral arterial disease11. This article delves into analyzing the usage patterns of one of the most commonly prescribed OADs in India glimepiride and metformin along with other OADs.
Material and Methods
Study Design
The study was a retrospective, multicenter, observational case-based questionnaire survey on T2DM patients undergoing pharmacotherapy.
It aimed to collect data on clinical utilization patterns of glimepiride/metformin FDC (fixed-dose combination) with other OADs, demographics, and comorbidities. Independent t-test was used to compare the change in fasting plasma glucose (FPG), postprandial plasma glucose (PPG), and HbA1c between two groups and Fisher’s exact and Chi-square tests were used to compare categorical variables. All the reported p-values were two-sided and p-values <0.05 were considered to indicate statistical significance. Statistical analysis was performed using SPSS® Version 23.0 software.
Study Population
Patients of both sexes, aged above 18 years, diagnosed with T2DM who received glimepiride/metformin and patients with comorbidities who were prescribed medications. T2DM patients below the age of 18 years and who were on monotherapy for T2DM were excluded from the study.
Data Collection
A case report format (CRF) was developed to determine the pattern of use of different strengths of glimepiride/metformin FDCs with or without other oral hypoglycemic agents in diabetes patients. Vital parameters including body mass index (BMI), hypertension, and other comorbidities, T2DM duration, dosage regimens of different OADs and the laboratory glycemic investigations were also included.
A questionnaire was sent to 500 healthcare professionals in India via an online portal for a descriptive analysis.
Data was collected digitally from clinicians through digitized CRF, clinical characteristics, laboratory findings, and treatment regimens from electronic medical records or doctor's records. The data was independently supervised by two investigators and reviewed by different investigators.
Statistical Analysis
All continuous variables were expressed as mean ± standard deviation (SD) or median with the interquartile range per the data distribution. Categorical variables were expressed as numbers and their respective percentage. Independent t-test was used to compare the change in FPG, PPG, and HbA1c between two groups and Fisher’s exact and Chi-square tests were used to compare categorical variables. All the reported p-values were two-sided and p-values <0.05 were considered to indicate statistical significance. All data entries and statistical analyses were performed by using SPSS@ Version 23.0 software.
Compliance with Ethics Guidelines
The study was approved by the ethical committee. All procedures adhered to the ethical standards established by the relevant institutional or national research committees. Since the study used an anonymized database and was done retrospectively, patient consent was not needed.
Results
The study analysis included responses from 500 HCPs. It showed that 6,250 patients received glimepiride/metformin FDC. The ages of the patients were between 18 to 90 years and a mean BMI was 27.97 ± 4.29 who received glimepiride/metformin combination. The mean (±SD) duration for which patients were having diabetes was 7.54 ± 3.48 years. The HbA1c was found to be 8.81% before treatment, which decreased to 7.75% after treatment. Among the 6,250 patients, 1,704 patients also received other OADs, where some patients received more than one OADs. Figure 2 shows the trend of OADs in combination with Metformin + Glimepiride.
DPP4i was prescribed the most (1,064 patients) followed by sodium-glucose cotransporter-2 inhibitors (SGLT2i) (573 patients), pioglitazone (229 patients), alpha-glucosidase inhibitor (AGI) (207 patients), insulin (178 patients), and lastly glucagon-like peptide 1 receptor agonist (GLP1RA) being prescribed in 35
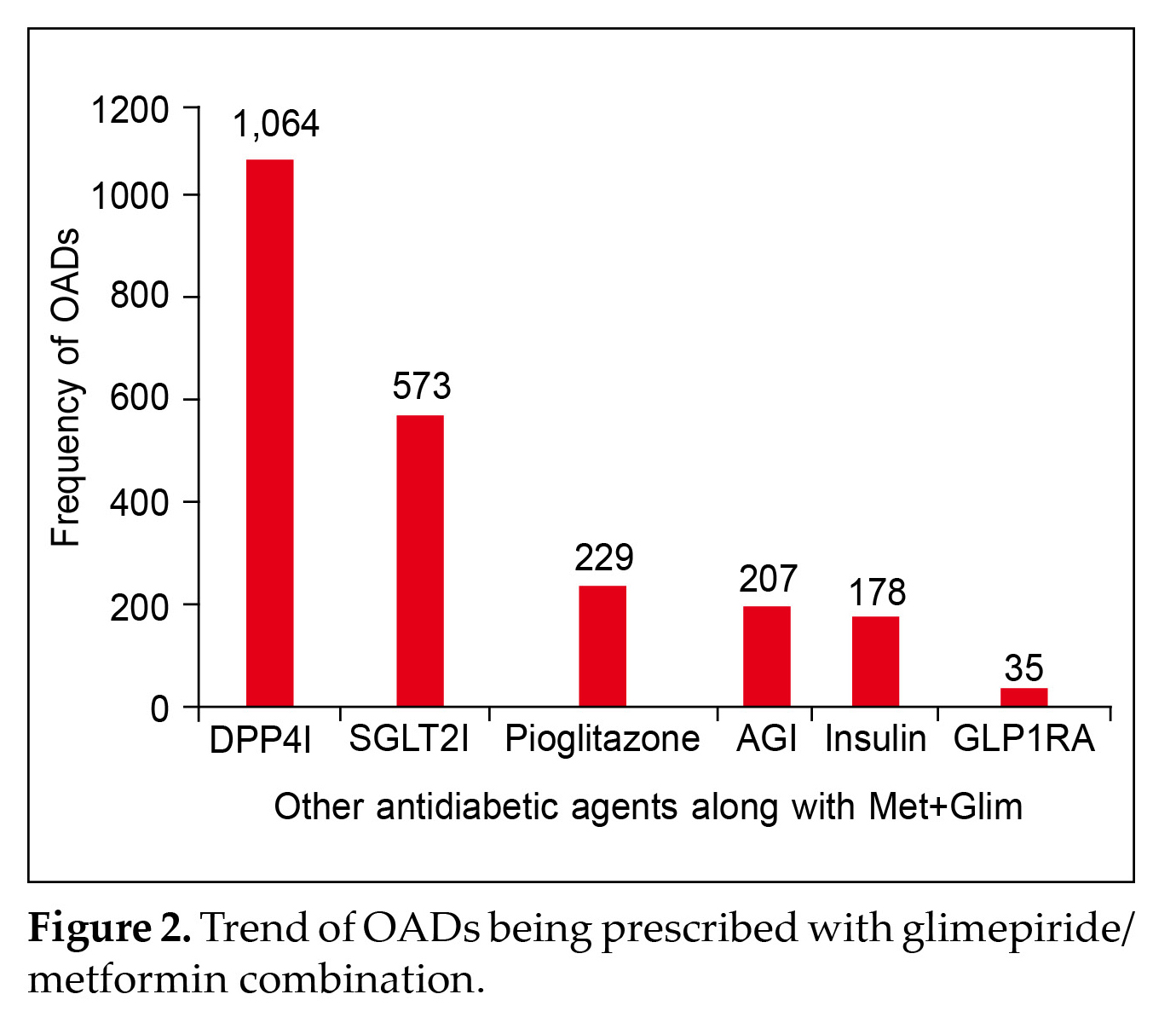
patients along with the combination as is depicted in Figure 2.
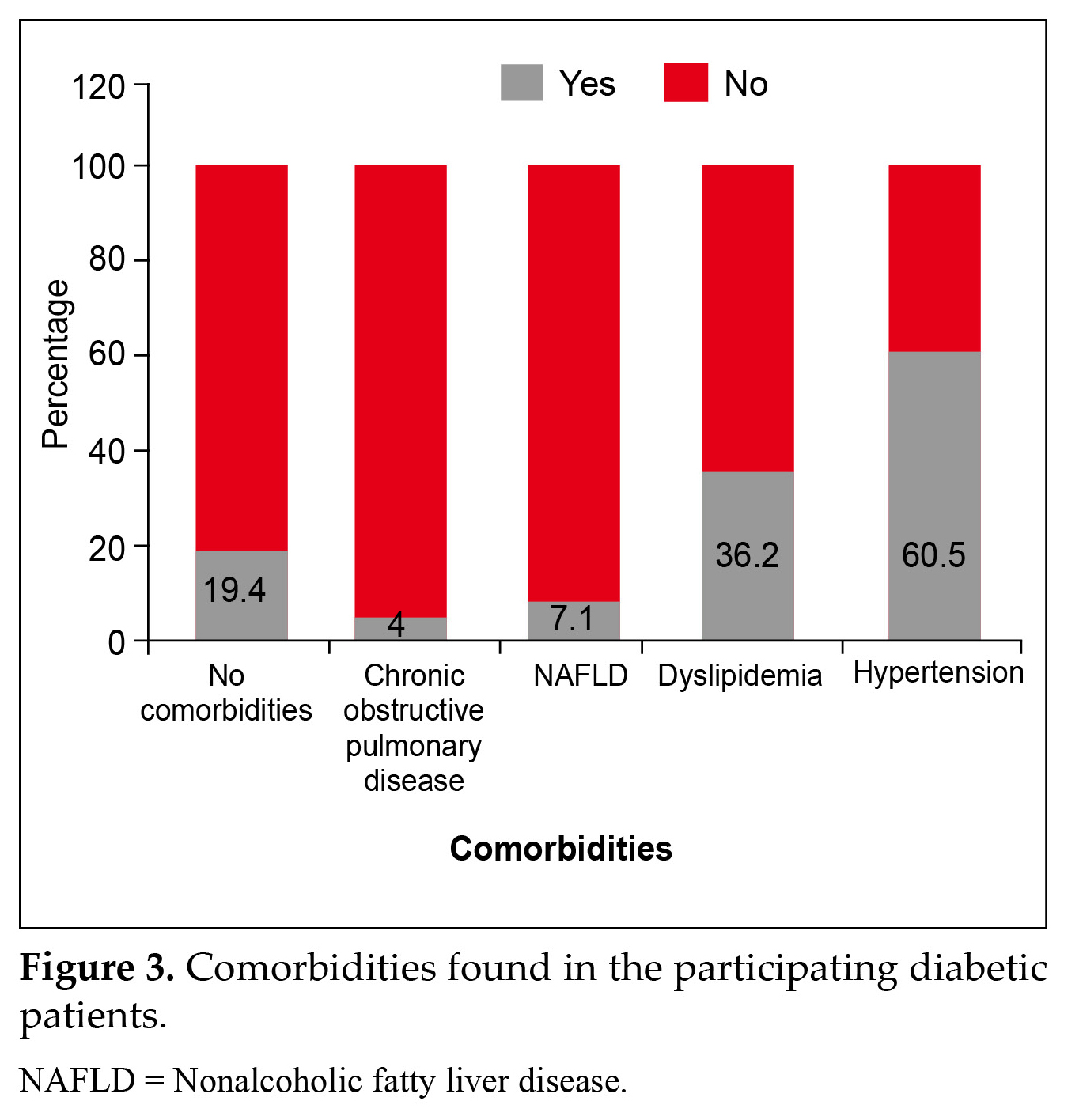
Hypoglycemia was observed in very few patients (4.49%). Hypertension was the most prevalent (60.5%) comorbidity in the studied patient population mostly aged between 18 to 90 years as is observed in Figure 3, followed by dyslipidemia (36.2%) among the patients included.
Discussion
Over half of the populations in India are at risk of having diabetes at some time in their life, which is making it a public health problem. An assessment in the review article published in 2021 stated that people residing in the cities and metropolitan regions in India are more likely to get diabetes due to lifestyle changes which increases the person’s BMI, a risk factor for diabetes. A significant surge is also being observed in rural parts of India3.
Hence, selection of proper drugs to control the rise becomes essential. Presently, there are approximately 60 medications that have been authorized by the Food and Drug Administration (FDA) as therapeutic choices for the treatment of T2DM11.
The selection of these drugs is typically affected by the numerous national and international recommendations created by various organizations in an effort to improve the management of diabetes mellitus12-14.
For many T2DM patients, combination therapy is necessary to maintain blood glucose levels within the desired range and prevent complications from diabetes. Some OADs are advantageous for heart and renal health as well as weight loss15,16.
Progressive beta-cell loss, a hallmark of T2DM, necessitates the sequential addition of various oral and injectable drugs to provide the best possible glycemic control. As the condition worsens, combination therapy becomes the need of the hour to establish appropriate glycemic control. Presence of comorbid conditions such as dyslipidemia, hypertension, and cardiovascular disease along with polypharmacy which comes with an increased load of pills and dose frequency, adds to the burden of medication17,18.
In the present study, hypertension was the most prevalent (60.5%) comorbidity in the total studied patient population, followed by dyslipidemia and nonalcoholic fatty liver disease (NAFLD).
It was further seen that among 1,704 patients (some of whom also received more than one OADs along with Metformin + Glimepiride combination), 1217 (71.04%) were hypertensive, 720 (42.22%) had dyslipidemia, 1,164 (68.31%) had lifestyle related risk factors. Hence, the presence of these comorbidities might be one of the causes to add other OADs.
Also studies have shown that one method to improve drug adherence is to use additional OADs along with the existing combination therapy. Drug combinations have been associated with improved compliance and improved glycemic control17.
The pattern observed in the present study showed that along with the glimepiride/metformin combination, DPP4i was prescribed the most (1,064 patients) followed by SGLT2i (573 patients), pioglitazone (229 patients), AGI (207 patients), insulin (178 patients), and lastly GLP1RA being prescribed in only 35 patients along with the combination as shown.
Dipeptidyl peptidase-4 (DPP-4) is a serine protease that cleaves and inactivates hormones, leading to decreased insulin secretion and disrupted visceral fat metabolism. It also plays a role in regulating postprandial glucose by degrading glucagon-like peptide 1 (GLP-1). DPP4i has been explored as a therapeutic target for the treatment and management of T2DM19. Research has demonstrated that DPP4i possess a favorable therapeutic profile, do not increase cardiovascular risk, and are safe and effective for most patients with T2DM20.
A large retrospective real-world investigation shows that adding a DPP4i to the existing medication improves glucose control in normal diabetic outpatient clinical practice. While DPP4i and gliclazide both increase endogenous insulin secretion, DPP4i has a stronger physiological effect that is meal-dependent and may be better able to enhance beta and alpha cell activity, which would lead to improved glycemic control21. DPP4i were found to have no hypoglycemia risks, neutral effect with respect to weight change, atherosclerotic cardiovascular disease (ASCVD) and renal diseases. Also it was found to decrease postprandial triglycerides and blood pressure (BP), hence is beneficial for hypertensive diabetic patients. In the current study, most of the patients taking the combination were also hypertensive and were also given DPP4i. This is in accordance with the fact that DPP4i helps in lowering (BP) and with blood glucose level22.
Metformin increases insulin sensitivity, while glimepiride increases - cell glucose sensitivity and promotes endogenous insulin production. A complementary mechanism of action between glimepiride and metformin results in a considerable decrease in glycemic indices (FPG, PPG, and HbA1c levels)10,23.
In India, SUs are second-line medications for T2DM patients who are not obese and also reduce the risk of hypoglycemia. Hence, SUs are preferred in this population24. Glimepiride is also a desirable choice for the management of people with long-term diabetes due to its shown cardiovascular safety/neutrality and decreased hypoglycemic episodes25. Hence, addition of other OADs like DPP4i along with the glimepiride/metformin seems to be beneficial in T2DM patients having comorbidities such as hypertension, dyslipidemia, etc.
In T2DM patients, strict glycemic management lowers the related comorbidities and raises quality of life26. According to the United Kingdom Prospective Diabetes Study (UKPDS) trial, there is a 12% to 43% reduction in the risk of diabetes-related mortality and morbidity for every 1% drop in HbA1c27,28.
Glimepiride increases cell sensitivity to glucose and promotes endogenous insulin production, whereas metformin increases sensitivity to insulin. Glycemic markers (FPG, PPG, and HbA1c levels) are significantly decreased when glimepiride and metformin are used in combination due to their complementary mechanisms of action.
When compared to older-generation SUs, glimepiride offers a number of benefits: weight-neutral effects, lack of cardiovascular risk, and fewer hypoglycemia episodes. It also has extrapancreatic effects, which are superior, enhanced insulin secretion22. Glimepiride + Metformin show synergistic effects by reducing hypoglycemia, weight gain, and cardiovascular risks, good glycemic control and improved safety profile23,29-31.
Prasanna Kumar et al also reported similar observations in a trial which showed that the combination had a good to outstanding effectiveness and tolerability in the majority of patients (97.3% and 96.6%)32.
Another prospective research found that diabetic individuals on glimepiride experienced fewer hypoglycemia episodes than those taking glibenclamide33.
Glimepiride’s documented cardiovascular safety/neutrality and reduced hypoglycemia episodes make it an attractive alternative for the management of persons with long-standing diabetes25. SUs are affordable and are also effective alternatives to other more recent antidiabetic drugs31.
Combination drugs in diabetes treatment are cost-effective as they reduce the need for multiple medications, simplify dosing, and improve patient adherence, ultimately lowering overall health care costs. Hence, the combinations are preferred in developing countries like India34. This study showed that among 6,250 patients in the age between 18 to 90 years and a mean BMI of 27.97 ± 4.29 received glimepiride/metformin combination.
The mean (±SD) duration for which patients were having diabetes was 7.54 ± 3.48 years. The HbA1c was found to be 8.81 ± 1.25 before treatment, which decreased to 7.75 ± 3.62 after treatment. The mean FPG values before treatment was 190.46 ± 53.20 which reduced to 139.50 ± 39.51 mg/dL after treatment, while the mean PPG values before treatment was 274.62 ± 32.11, which decreased to 165.22 ± 45.63 mg/dL after treatment.
Among the 6,250 patients, 1,704 patients also received other OADs, 758 achieved HbA1c <7. Moreover, 228 patients achieved target FPG values, i.e., FPG values <100 and 95 achieved target PPG values, i.e., values <125.
In the current study, a significant decrease in the HbA1c, FPG, and PPG was observed, which is in similar lines with the findings by Hassan and Abd-Allah (2015), Surendra Kumar (2021), Shrivastava et al (2023)23,30,35.
A variety of antidiabetic medications are now used as monotherapy or in combination for treating T2DM. Several studies have shown that in various Afro-Asian nations, including India, modern SUs alone or in combination with metformin are the OADs prescribed most often as they achieved better HbA1c, FPG, and PPG when used along with different OADs36,37.
Conclusion
Prevalence of diabetes is increasing in India. Glimepiride/Metformin FDC can be used with various other OADs for better management of diabetes among patients with additional comorbidities. The study shows that DPP4i was the most common OAD being prescribed along with glimepiride/metformin combination.
This new age approach offers valuable insights into the multifaceted management of diabetes, highlighting the importance of individualized treatment strategies for Indian patients. The study would help HCPs to understand and optimize diabetes management in a better manner, which in turn would enable patients to lead healthier and more productive lives.
Acknowledgment
We acknowledge the support provided by the entire team PMT for their support during the conduct of this study. The medical writing support was provided by the IJCP Team.
Financial Support and Sponsorship
This study was funded by USV Private Limited, Mumbai, Maharashtra, India.
Conflicts of Interest
Dr Aushili M, Dr Ashish Prasad, Dr Abhijit Pednekar are employees of USV Private Limited, Mumbai, Maharashtra, India.
References
- Microvascular Complications and Foot Care: Standards of Medical Care in Diabetes-2021. Diabetes care. 2021;44(Suppl 1):S151-s67.
- Kumar A, Gangwar R, Zargar AA, Kumar R, Sharma A. Prevalence of diabetes in India: A review of IDF Diabetes Atlas 10th edition. Curr Diabetes Rev. 2024;20(1):e130423215752.
- Pradeepa R, Mohan V. Epidemiology of type 2 diabetes in India. Indian J Ophthalmol. 2021;69(11):2932-8.
- Sahay RK, Mittal V, Gopal GR, Kota S, Goyal G, Abhyankar M, et al. Glimepiride and metformin combinations in diabetes comorbidities and complications: real-world evidence. Cureus. 2020;12(9):e10700.
- Padhi S, Nayak AK, Behera A. Type II diabetes mellitus: a review on recent drug based therapeutics. Biomed Pharmacother. 2020;131:110708.
- Kalra S, Aamir AH, Raza A, Das AK, Azad Khan AK, Shrestha D, et al. Place of sulfonylureas in the management of type 2 diabetes mellitus in South Asia: a consensus statement. Indian J Endocrinol Metab. 2015;19(5):577-96.
- IDF Clinical Practice Recommendations for Managing Type 2 Diabetes in Primary Care. 2017. Available at: https:// www.idf.org/e-library/guidelines/128-idf-clinical-practicerecommendations-for-managing-type-2-diabetes-inprimary-care.html. Accessed September 12, 2024.
- RSSDI clinical practice recommendations for the management of type 2 diabetes mellitus 2022. Int J Diabetes Dev Ctries. 2022;42(1):1-143.
- WHO Guidelines Approved by the Guidelines Review Committee. Guidelines on second-and third-line medicines and type of insulin for the control of blood glucose levels in non-pregnant adults with diabetes mellitus. Geneva: World Health Organization; 2018.
- Basit A, Riaz M, Fawwad A. Glimepiride: evidence-based facts, trends, and observations (GIFTS). [Corrected]. Vasc Health Risk Manag. 2012;8:463-72.
- Rosenstock J, Kahn SE, Johansen OE, Zinman B, Espeland MA, Woerle HJ, et al; CAROLINA Investigators. Effect of linagliptin vs glimepiride on major adverse cardiovascular outcomes in patients with type 2 diabetes: the CAROLINA Randomized Clinical Trial. JAMA. 2019;322(12):1155-66.
- Mohan V, Saboo B, Khader J, Modi KD, Jindal S, Wangnoo SK, et al. Position of sulfonylureas in the current ERA: review of national and international guidelines. Clin Med Insights Endocrinol Diabetes. 2022;15:11795514221074663.
- Davies MJ, D'Alessio DA, Fradkin J, Kernan WN, Mathieu C, Mingrone G, et al. Management of Hyperglycemia in Type 2 Diabetes, 2018. A Consensus Report by the American Diabetes Association (ADA) and the European Association for the Study of Diabetes (EASD). Diabetes Care. 2018;41(12):2669-701.
- American Diabetes Association 9. Pharmacologic Approaches to Glycemic Treatment: Standards of Care in Diabetes-2024. Diabetes care. 2024;47(Suppl 1):S158-S78.
- Milligan S. Combination therapy for the improvement of long-term macrovascular and microvascular outcomes in type 2 diabetes: Rationale and evidence for early initiation. J Diabetes Complications. 2016;30(6):1177-85.
- Nawaz S, Chinnadurai R, Al-Chalabi S, Evans P, Kalra PA, Syed AA, et al. Obesity and chronic kidney disease: a current review. Obes Sci Pract. 2023;9(2):61-74.
- John M, Gopinath D, Kalra S. Triple fixed drug combinations in type 2 diabetes. Indian J Endocrinol Metab. 2015;19(3):311-3.
- Donnan PT, MacDonald TM, Morris AD. Adherence to prescribed oral hypoglycaemic medication in a population of patients with type 2 diabetes: a retrospective cohort study. Diabet Med. 2002;19(4):279-84.
- Yin R, Xu Y, Wang X, Yang L, Zhao D. Role of dipeptidyl peptidase 4 inhibitors in antidiabetic treatment. Molecules. 2022;27(10):3055.
- Deacon CF. Dipeptidyl peptidase 4 inhibitors in the treatment of type 2 diabetes mellitus. Nat Rev Endocrinol. 2020;16(11):642-53.
- Fadini GP, Bottigliengo D, D’Angelo F, Cavalot F, Bossi AC, Zatti G, et al. Comparative effectiveness of DPP-4 inhibitors versus sulfonylurea for the treatment of type 2 diabetes in routine clinical practice: a retrospective multicenter real-world study. Diabetes Ther. 2018;9(4):1477-90.
- Feingold KR. Oral and Injectable (Non-Insulin) Pharmacological Agents for the Treatment of Type 2 Diabetes. [Updated 2022 Aug 26]. In: Feingold KR, Anawalt B, Blackman MR, et al., editors. Endotext [Internet]. South Dartmouth (MA): MDText.com, Inc.; 2000-. Available from: https://www.ncbi.nlm.nih.gov/books/NBK279141/
- Shrivastava A, Kesavadev J, Mohan V, Saboo B, Shrestha D, Maheshwari A, et al. Clinical evidence and practice-based guidelines on the utility of basal insulin combined oral therapy (metformin and glimepiride) in the current era. Curr Diabetes Rev. 2023;19(8):e090123212444.
- RSSDI Clinical Practice Recommendations for the Management of Type 2 Diabetes Mellitus 2022. Int J Diabetes Dev Ctries.
- ElSayed NA, Aleppo G, Aroda VR, Bannuru RR, Brown FM, Bruemmer D, et al; American Diabetes Association. 9. Pharmacologic Approaches to Glycemic Treatment: Standards of Care in Diabetes—2023. Diabetes Care. 2023;46(Suppl 1):S140-57.
- Rodríguez-Gutiérrez R, Montori VM. Glycemic control for patients with type 2 diabetes mellitus: our evolving faith in the face of evidence. Circ Cardiovasc Qual Outcomes. 2016;9(5):504-12.
- Intensive blood-glucose control with sulphonylureas or insulin compared with conventional treatment and risk of complications in patients with type 2 diabetes (UKPDS 33). UK Prospective Diabetes Study (UKPDS) Group. Lancet. 1998;352(9131):837-53.
- Lind M, Imberg H, Coleman RL, Nerman O, Holman RR. Historical HbA1c values may explain the type 2 diabetes legacy effect: UKPDS 88. Diabetes Care. 2021;44(10):2231-7.
- Das AK, Saboo B, Chawla R, Aravind SR, Rajput R, Singh AK, et al. Time to reposition sulfonylureas in type 2 diabetes management in Indian context: A pragmatic practical approach. Int J Diabetes Dev Ctries. 2023:1-19.
- Hassan M, Abd-Allah GM. Effects of metformin plus gliclazide versus metformin plus glimepiride on cardiovascular risk factors in patients with type 2 diabetes mellitus. Pak J Pharm Sci. 2015;28(5):1723-30.
- Kalra S, Das AK, Baruah MP, Unnikrishnan AG, Dasgupta A, Shah P, et al. Glucocrinology of modern sulfonylureas: clinical evidence and practice-based opinion from an international expert group. Diabetes Ther. 2019;10(5):1577-93.
- Prasanna Kumar KM, Seshadri K, Aravind SR, Deb P, Modi KD, Gopal RA, et al. Real-world observational study of glimepiride and metformin fixed-dose combination along with insulin in the management of type 2 diabetes mellitus: Indian experience. Cureus. 2021;13(1):e13020.
- Zekry R, Omran GA, El-Gharbawy NM, Werida RH. Comparative study of Dapagliflozin versus Glimepiride effect on insulin regulated aminopeptidase (IRAP) and interleukin-34 (IL-34) in patient with type 2 diabetes mellitus. Sci Rep. 2023;13(1):6302.
- Leichter SB, Thomas S. Combination medications in diabetes care: an opportunity that merits more attention. Clin Diabetes. 2003;21(4):175-8.
- Kumar S. Comparison of safety and efficacy of glimepiride-metformin and vildagliptin-metformin treatment in newly diagnosed type 2 diabetic patients. Indian J Endocrinol Metab. 2021;25(4):326-31.
- Kalra S, Bahendeka S, Sahay R, Ghosh S, Md F, Orabi A, et al. Consensus recommendations on sulfonylurea and sulfonylurea combinations in the management of type 2 diabetes mellitus - International Task Force. Indian J Endocrinol Metab. 2018;22(1):132-57.
- Mandal S, Maiti T, Das AK, Das A, Mandal A, Sarkar BS,
et al. Drug utilization study in patients with type 2 diabetes mellitus attending diabetes clinic of a tertiary care hospital in rural Bengal. Int J Basic Clin Pharmacol. 2016;5(4):1647-54.